INTRODUCTION
MATERIALS AND METHOD
Plant materials collection and classification
Field cultivation and data recording
Data analysis
RESULTS AND DISCUSSION
Variations of qualitative agronomic characteristics
Variations of quantitative agronomic characteristics
Hierarchical cluster (HCA), Principal component (PCA), and correlation analyses
INTRODUCTION
Adzuki bean (Vigna angularis (Willd.) Ohwi & Ohashi) is one of the most important legumes in Asian countries. Although the plant is grown in other countries such as the United States and Canada, it is primarily cultivated in Korea, Japan, and China (Johnson et al., 2022; Tayade et al., 2022). In these countries, adzuki bean seeds are majorly consumed as soup and paste (Li-xia et al., 2021; Takahama et al., 2016). Besides, they are cooked and consumed with other common foods such as rice (Takahama et al., 2019). Adzuki bean seeds are rich sources of nutritional and other health-promoting metabolites including minerals, carbohydrates, fatty acids, vitamins, protein, fibers, flavonoids, anthocyanins, and saponins, among others (Ha et al., 2021; Jeong et al., 2021; Yadav et al., 2018). Moreover, adzuki bean seed extracts and isolates are known to exhibit several pharmacological properties such as antioxidant, antiobesity, antidiabetic, and anticancer properties (Aslinah et al., 2018; Li et al., 2022; Zhao et al., 2021). Owing to these properties, adzuki bean seeds are becoming a research focus in recent years although multidisciplinary studies focusing on adzuki bean genetic materials are still scarce (Srivastava et al., 2018).
The analysis of genetic diversity among large populations of adzuki bean genetic materials is important for their conservation, selection, and utilization during the development of improved cultivars (Li et al., 2020a; Wang et al., 2019). Compared with other legumes, however, the diversity among adzuki bean genotypes is considered to be lower and hence, success stories regarding the breeding of improved adzuki bean cultivars are infrequent. On the other hand, several environmental and genetic factors including growing location, year of cultivation, temperature, genotype, and disease, among others, are affecting not only the agro-morphological features of adzuki beans but also their field production (Dong et al., 2016; Han et al., 2021; Tripathi & Kim, 2022). Because of these, the improvement of adzuki bean cultivars resistant to diseases and environmental stresses has become one of the research objectives in recent years (Chun et al., 2021; Keneni et al., 2011; Li et al., 2020b). To assist the development of improved adzuki bean cultivars, studies that investigate a large population of adzuki bean genetic materials are continuously recommended and are of great importance since they provide a much better view of the existing genetic diversity (Kang et al., 2015; Pandiyan et al., 2021; Wang et al., 2019).
Agro-morphological traits are the primary phenotypic data used to assess the diversity among plant genetic materials and document the physical appearances, field performances, and quality traits of plant genotypes. They are also important factors influencing their selection and utilization by consumers, farmers, and breeders (Ammar et al., 2015). Previously, few studies investigated the genetic diversity of adzuki bean genetic materials grown in Korea using agro-morphological features and the majority of these studies relied on locally collected genetic resources (Rho et al., 2003; Tayade et al., 2022; Yoon et al., 2012). As far as we could tell, there are no surplus studies that investigated the genetic diversity of adzuki beans of different origins grown in Korea. In this study, we collected 252 adzuki bean germplasms originating from China, Japan, and Korea and cultivated them recently in Korea, and assessed their genetic diversity using eighteen agro-morphological features as well as in comparison to three widely known Korean adzuki bean cultivars including Geomguseul, Arari, and Chungjupat. The results of this study could be useful in determining the adaptability of adzuki bean genetic materials from various origins in Korea, as well as facilitating the use of adzuki beans in breeding programs.
MATERIALS AND METHOD
Plant materials collection and classification
The seeds of 252 adzuki bean genetic materials were obtained from the gene bank of the National Agrobiodiversity Center, Rural Development Administration (RDA, Jeonju, Korea). The adzuki beans were classified according to their origin as Japan (n = 58), China (n = 99), and Korea (95). Among all the adzuki beans, 9 were cultivars, 17 were breeding lines, and the remaining 226 genotypes were landraces (Supplementary Table S1). Three widely known Korean adzuki bean cultivars including Geomguseul (black seed coat), Arari (red seed coat), and Chungjupat (red seed coat) were also obtained from the same source and used as control cultivars (Han et al., 2022).
Field cultivation and data recording
Field cultivation of all the adzuki bean seeds was conducted between June and October of 2021 in an experimental farm located at the National Agrobiodiversity Center, RDA, Jeonju, Korea (latitude/longitude: 35°49´38.37´´N/127°09´07.78´´E). On June 22, 2021, twenty seeds were sown in 90 cm apart rows with two seeds per hole at a 15 cm spacing for each adzuki bean. The accessions were grown under uniform weather and treatment conditions. Cultivation was conducted on clay loam soil and NPK fertilizer at a ratio of 3:3:3.4 kg a-1 was applied. The growth period lasted until October of the same year, and the average monthly temperature in the cultivation area was 23.0, 27.2, 25.9, 22.9, and 16.4°C, while the average monthly precipitation was 145.3, 255.1, 454.8, 162.1, and 37.0 mm in June, July, August, September, and October, respectively. Matured seeds were hand-harvested and a total of eighteen agro-morphological traits including ten qualitative characters (growth habit, growth type, hypocotyl color, leaf shape, flower color, degree of pubescence, pod color, seed shape, seed coat color, and seed coat lust) and eight quantitative traits (Rate of germination (RG), days to flowering (DF), days to maturity (DM), days from flowering to maturity (DFM), days to 50% flowering (D50F), number of pods per plant (PPP), number of seeds per pod (SPP), and one-hundred seeds weight (HSW)) were recorded from field performances and laboratory inspection. The rate of germination was calculated as a percentage by dividing the number of seeds germinated by the total number of seeds sown (Song et al., 2016). Similarly, the other agronomic traits were determined using the Hu et al. (2022) method.
Data analysis
Data were recorded from the field as well as laboratory inspections and values were reported as mean ± standard deviation (SD). Using xlstat software (Addinsfot, NY, USA), the difference between means was statistically evaluated by analysis of variance (ANOVA) at p < 0.05 level. Multivariate analyses including principal component (PCA) and hierarchical cluster (HCA) analyses were conducted using R-software version 4.0 (www.r-project.org) and used to view the distribution and associations of the adzuki bean genotypes and assess the variables that caused the most variation. Pearson’s correlation matrix was also generated using R-software and used to further view the relationship between variables.
RESULTS AND DISCUSSION
Variations of qualitative agronomic characteristics
Despite the high metabolite contents and nutritional values, adzuki beans are one of the most underutilized legumes and have only recently gained popularity. To maximize their production and application in food sectors, multidisciplinary studies that exploit their potential are constantly required (Bhatt et al., 2021; Chapman et al., 2022). Qualitative characters are important parameters to document the morphology and physical appearances of plant genotypes which in turn affect their preference and selection by consumers, farmers, and breeders (Rashid et al., 2020). Previous studies conducted in Korea and elsewhere showed wide differences in qualitative agronomical traits between adzuki bean genetic resources and other legumes including mung beans and soybeans, among others (Ammar et al., 2015; Rho et al., 2003; Yoon et al., 2012). Many of these studies also verified the influences of genotype, growth condition, and weather on the agro-morphological features of legumes. In this study, a total of ten qualitative agronomical characters were recorded for all the accessions as described before. Information regarding each of the 252 adzuki bean genetic materials, their introduction number, and qualitative agronomical properties can be viewed in Supplementary Table S1. The frequency distribution of the accessions across each character is summarized in Table 1. The adzuki beans showed wide variations in some of the most important agronomic traits. Similar to all the control cultivars, the majority of them showed an upright growth habit (f = 236) and a semi-determinate growth type (f = 247) regardless of their origin. The adzuki beans also developed either green or purple hypocotyl colors, the former being the most dominant in Chinese (f = 96), Japanese (f = 55), and Korean (f = 90) adzuki beans. Other traits that showed wide differences include leaf shape, flower color, pod color, and seed coat color. Xiphoid with a wide base was the dominant leaf shape in Japanese adzuki beans compared to Korean and Chinese adzuki beans where xiphoid and wide elliptical shapes dominated, respectively. The majority of the adzuki beans developed yellow flower color (f = 226) like the control cultivars. The majority of Chinese adzuki beans tend to possess a medium degree of pubescence, while the majority of Korean and Japanese adzuki beans had plenty and a medium degree of pubescence, respectively. Besides, 44 accessions from Japan, 70 accessions from China, and 65 accessions from Korea developed elliptical seeds, while the remaining were either long or short cylindrical. Many of the Chinese (f = 78), Japanese (f = 48) and Korean (f = 79) adzuki beans developed a red-seed coat. Though not frequent, other seed coat colors observed include gray (f = 26), Ivory (f = 4), olive (f = 2) and white (f = 6). In adzuki beans, seed coat color is considered to be one of the most important quality traits since it is closely associated with the biosynthesis pathways of disease-deterring metabolites (Chu et al., 2021; Horiuchi et al., 2015). Therefore, the development of several seed coat colors in the adzuki beans could be a good input to view their diversity in terms of metabolite compositions. Moreover, the observed variations in other characters could provide a wide spectrum of options to select typical genotypes during the development of improved cultivars.
Table 1.
Frequency (f) and percentage (%) distribution of qualitative agronomical traits in 252 adzuki bean germplasms of different origins cultivated in Korea.
Variations of quantitative agronomic characteristics
Quantitative agronomical traits are also determinant factors during the selection of important genotypes (Hu et al., 2022). In our study, a total of eight quantitative traits mostly related to growth and yield were documented and the adzuki beans showed wide variations (Supplementary Table S2). Fig. 1 depicts the distribution pattern of each quantitative trait, while Table 2 summarizes the numerical and statistical values. The largest degree of variation was observed in PPP (CV ≥ 41.16%) followed by HSW (CV ≥ 24.39%) and SPP (CV ≥14.37%) regardless of origin. In contrast, the adzuki beans showed the least variation in their DM (CV ≤ 6.48%). The skewness of RG and PPP was far from zero indicating asymmetric distributions of the accessions. Except for DF, D50%F, and SPP, the distribution pattern of the remaining variables showed positive kurtosis (Table 2) signifying the presence of outlier accessions (Fig. S1).
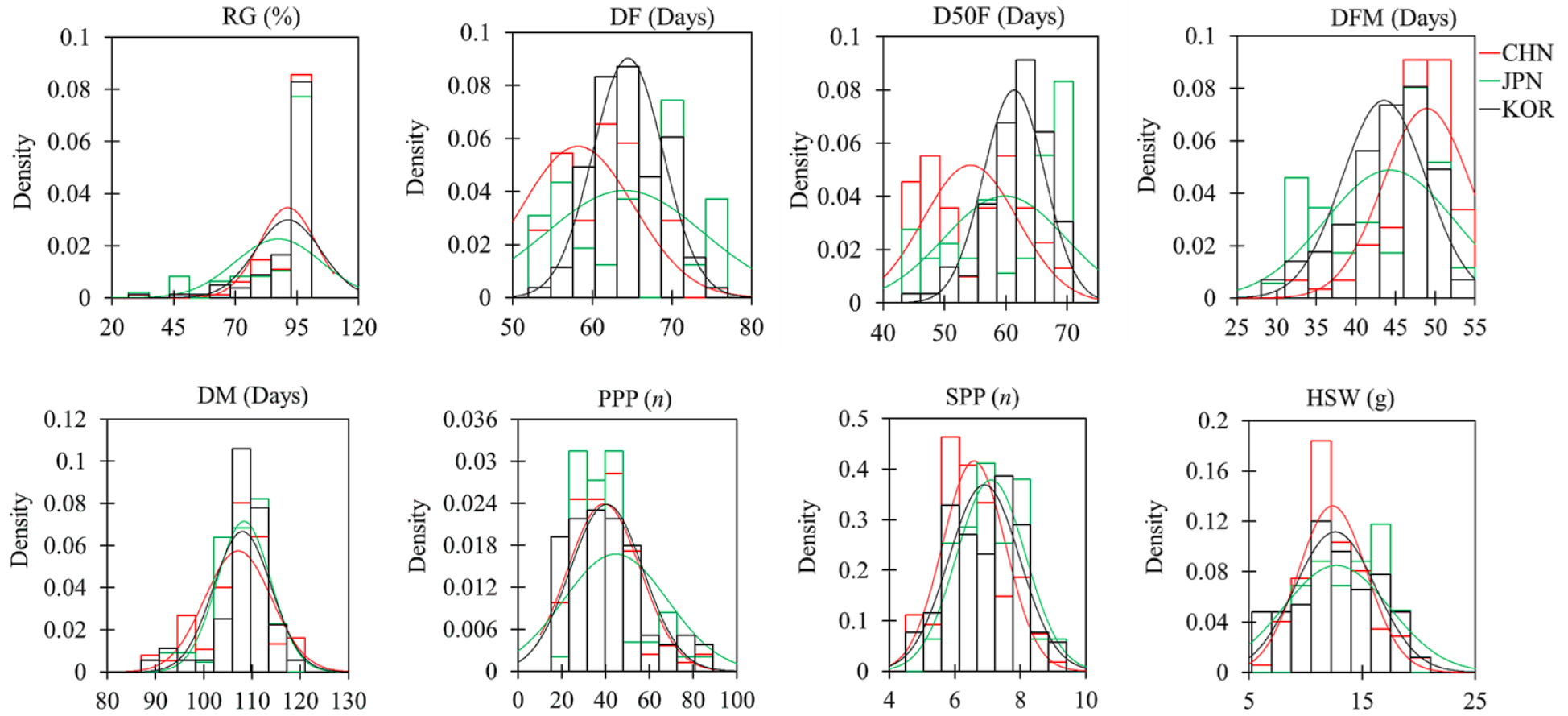
Fig. 1
Distribution of quantitative agronomical characters in 252 adzuki beans according to their origin. CHN: China, DF: Days to flowering, DFM: Days from flowering to maturity, DM: Days to maturity, D50F: Days to 50% flowering, RG: Rate of germination, HSW: One-hundred seeds weight, JPN: Japan, KOR: Korea, PPP: Pods per plant, SPP: Seeds per pod.
Table 2.
Statistical data of the quantitative agronomical characteristics of 252 adzuki bean germplasms of different origins cultivated in Korea.
| Variable | Value | Origin | Total | Controls | Skewness | Kurtosis | ||||
| China | Japan | Korea | Geomguseul | Arari | Chungjupat | |||||
| RG (%) | Min | 26.67 | 26.67 | 26.67 | 26.67 | 100.00 | 98.00 | 95.00 | -2.28 | 5.74 |
| Max | 100.00 | 100.00 | 100.00 | 100.00 | ||||||
| Mean ± SD | 91.31 ± 11.47 a | 87.47 ± 17.51 a | 91.44 ± 13.29 a | 90.48 ± 13.84 | ||||||
| CV (%) | 12.62 | 20.19 | 14.53 | 15.29 | ||||||
| D50F (Days) | Min | 42.00 | 36.00 | 43.00 | 36.00 | 57.00 | 59.00 | 70.00 | -0.43 | -60 |
| Max | 70.00 | 73.00 | 70.00 | 73.00 | ||||||
| Mean ± SD | 54.23 ± 7.67 b | 59.84 ± 9.58 a | 61.36 ± 4.96 a | 58.21 ± 8.08 | ||||||
| CV (%) | 14.21 | 16.61 | 8.09 | 8.08 | ||||||
| DF (Days) | Min | 47.00 | 47.00 | 52.00 | 47.00 | 59.00 | 62.00 | 70.00 | -0.18 | -0.49 |
| Max | 76.00 | 77.00 | 76.00 | 77.00 | ||||||
| Mean ± SD | 58.22 ± 6.96 b | 64.09 ± 9.79 a | 64.49 ± 4.40 a | 61.94 ± 7.57 | ||||||
| CV (%) | 12.01 | 15.42 | 6.82 | 12.23 | ||||||
| DFM (Days) | Min | 31.00 | 28.00 | 28.00 | 28.00 | 49.00 | 32.00 | 36.00 | -0.37 | 0.07 |
| Max | 61.00 | 61.00 | 54.00 | 61.00 | ||||||
| Mean ± SD | 48.91 ± 5.49 a | 44.26 ± 8.10 b | 43.53 ± 5.27 b | 45.81 ± 6.61 | ||||||
| CV (%) | 11.28 | 18.46 | 12.10 | 14.43 | ||||||
| DM (Days) | Min | 87.00 | 94.00 | 87.00 | 87.00 | 108.00 | 94.00 | 106.00 | -0.95 | 1.70 |
| Max | 120.00 | 127.00 | 120.00 | 127.00 | ||||||
| Mean ± SD | 107.13 ± 6.91 a | 108.34 ± 5.54 a | 108.02 ± 5.96 a | 107.75 ± 6.28 | ||||||
| CV (%) | 6.48 | 5.15 | 5.52 | 5.83 | ||||||
| PPP (Number) | Min | 7.67 | 9.67 | 15.00 | 7.67 | 39.30 | 24.30 | 50.30 | 1.78 | 5.73 |
| Max | 116.00 | 143.67 | 88.00 | 143.67 | ||||||
| Mean ± SD | 39.25 ± 16.60 a | 44.54 ± 23.62 a | 40.45 ± 16.65 a | 40.92 ± 18.58 | ||||||
| CV (%) | 42.50 | 53.49 | 41.16 | 45.41 | ||||||
| SPP (Number) | Min | 4.50 | 4.40 | 4.40 | 4.40 | 8.10 | 8.60 | 8.10 | 0.12 | -0.44 |
| Max | 8.90 | 9.80 | 9.80 | 9.80 | ||||||
| Mean ± SD | 6.58 ± 0.95b | 7.11 ± 1.04 a | 7.11 ± 1.08 a | 6.82 ± 1.04 | ||||||
| CV (%) | 14.57 | 14.82 | 14.69 | 15.29 | ||||||
| HSW (g) | Min | 4.43 | 2.30 | 5.23 | 2.30 | 11.90 | 15.90 | 17.80 | -0.28 | 0.13 |
| Max | 20.37 | 21.47 | 20.03 | 21.47 | ||||||
| Mean ± SD | 12.37 ± 3.00 a | 12.72 ± 4.66 a | 12.61 ± 3.56 a | 12.54 ± 3.65 | ||||||
| CV (%) | 24.39 | 36.96 | 28.20 | 29.11 | ||||||
The variations of each were statistically analyzed. RG was in the range of 26.67~100.00%. By comparison, Korean adzuki beans (91.44%) showed a greater average RG followed by Chinese (91.31%) and Japanese (87.47%) adzuki beans although the variation was not significant (p < 0.05). Overall, the adzuki beans showed a high RG regardless of their origin signifying the adaptability of the majority of the genetic materials in the Korean environment (Redden et al. 2009). Likewise, the D50F, DF, and DM were in the ranges of 36~73, 47-~77, and 87~127 days, respectively. Accessions IT275744 also took the shortest days to flower together with accessions IT280303 and IT293151 (from China), and accessions IT238547, IT270036, IT215376, IT294649, and IT236590 (from Japan). Likewise, accessions IT275744 and IT236567 from China and IT186272 and IT208937 from Korea were the earliest to mature followed by another Chinese accession, IT236596. In contrast, the Japanese accession IT267508 was the latest to mature in 127 days followed by four Chinese accessions (IT280291, IT280293, IT280298, and IT280296) and a Korean accession (IT296742) each taking 120 days (Supplementary Table S2). Based on origin, the average D50F, DF, and DM were shorter in Chinese adzuki beans, the former two being significantly different from those of Korean and Japanese adzuki beans (p < 0.05) (Table 2). Previous studies also observed variations in the DF and DM, and some of these findings were comparable with our observations. For instance, Rho et al. (2003) studied 361 adzuki bean genotypes grown in Korea and observed DF and DM in the ranges of 47~80 and 73~128 days, respectively which agreed with our results. In another study, Yoon et al. (2012) reported much wider values for DF (51~110 days) and DM (81~150 days) in 150 adzuki bean germplasms cultivated in Korea. Redden et al. (2009) also found varied DF and DM in 231 Chinese adzuki bean germplasms cultivated in different provinces. In addition to genetic variations, differences in the cultivation year, place of cultivation, weather conditions, and exposure to diseases, among others could cause such disparities (Hu et al., 2022). Among the control cultivars, Geomguseul was the earliest to flower (in 59 days) while Arari was the fastest to mature (in 94 days). Among the 252 accessions, 29.76% of the accessions were early flowering (< 59 days) than all the three control cultivars while only 3.97% were early maturing (< 94 days). The development of early maturing legume genotypes is one of the primary objectives in breeding (Ammar et al., 2015). Accordingly, a total of ten early maturing accessions including 5 accessions from China (IT236567 and IT275744 (87 days), IT236596 (90 days), IT293151 and IT280303 (93 days)) and other five accessions from Korea (IT186272 and IT208937 (87 days), IT168012 and IT168010 (each 91 days), and IT208913 (92 days)) were identified as early maturing and hence, could be vital resources (Table 3).
Table 3.
Characteristics of the top ten high PPP-yielding and early maturing adzuki bean accessions.
| High PPP-yielding adzuki beans | Early maturing adzuki beans | ||||||||||||||
| IT No. | PPP (n) | Genotype | Origin | Size | DF (Days) | DM (Days) | SPP (n) | IT No. | DM (Days) | Genotype | Origin | Size | DF (Days) | PPP (n) | SPP (n) |
| 216289 | 143.7 | Landrace | Japan | Small | 76 | 108 | 8.5 | 236567 | 87 | Landrace | China | Small | 51 | 35.3 | 7.8 |
| 216304 | 127.0 | Landrace | Japan | Small | 76 | 108 | 7.6 | 275744 | 87 | Landrace | China | Small | 47 | 11.7 | 6.8 |
| 280307 | 116.0 | Landrace | China | Medium | 59 | 108 | 8.6 | 236596 | 90 | Landrace | China | Small | 48 | 40.7 | 7.7 |
| 216288 | 97.0 | Landrace | Japan | Medium | 77 | 112 | 7.9 | 280303 | 93 | Landrace | China | Small | 47 | 31.3 | 5.4 |
| 100898 | 88.0 | Landrace | Korea | Small | 59 | 94 | 6.5 | 293151 | 93 | Landrace | China | Small | 47 | 30.3 | 5.6 |
| 280296 | 85.7 | Landrace | China | Small | 65 | 120 | 6.6 | 208913 | 92 | Landrace | Korea | Medium | 62 | 37.3 | 6.0 |
| 216291 | 84.0 | Landrace | Japan | Small | 76 | 109 | 9.8 | 168010 | 91 | Landrace | Korea | Small | 58 | 24.3 | 4.8 |
| 280311 | 83.0 | Landrace | China | Small | 70 | 112 | 8.6 | 168012 | 91 | Landrace | Korea | Small | 55 | 17.3 | 6.6 |
| 208944 | 82.7 | Landrace | Korea | Small | 76 | 108 | 6.5 | 186272 | 87 | Landrace | Korea | Small | 52 | 32.0 | 7.5 |
| 296724 | 81.7 | Landrace | Korea | Medium | 65 | 109 | 7.6 | 208937 | 87 | Landrace | Korea | Small | 59 | 18.3 | 5.9 |
| Geomguseul | 39.3 | Cultivar | Korea | Small | 59 | 108 | 8.1 | Geomguseul | 108 | Cultivar | Korea | Small | 59 | 39.3 | 8.1 |
| Arari | 24.3 | Cultivar | Korea | Medium | 62 | 94 | 8.6 | Arari | 94 | Cultivar | Korea | Medium | 62 | 24.3 | 8.6 |
| Chungjupat | 50.3 | Cultivar | Korea | Medium | 70 | 106 | 8.1 | Chungjupat | 106 | Cultivar | Korea | Medium | 70 | 50.3 | 8.1 |
SPP (n), PPP (n), and HSW (g) are three of the most important yield-related quantitative traits (Gerrano & Thungo, 2022), and their values were 4.40~9.80, 7.67~143.67, and 2.30~21.47, respectively (Table 2, Fig. 1), with the observed values agreeing with previous studies (Rho et al., 2003; Yoon et al., 2012). Accession IT216289 produced the highest PPP followed by accessions IT216304 (n = 127.0), IT280307 (n = 116.0), and IT216288 (n = 97.0), all except IT216288 being from Japan (Supplementary Table S2). The lowest PPP was recorded for accession IT293155 followed by accessions IT294673 (n = 9.7), IT286521 (n = 11.3), and IT275744 (n = 11.7), each except IT294673 being from China. Accession IT216291, from Japan, had the highest SPP followed by other two Japanese accessions IT294643 and IT216294 (each with SPP of 9.1), and two Korean accessions IT104969 and IT296730 (each with SPP of 9.1). The lowest SPP was also recorded in a Japanese accession, IT294656, followed by a Korean accession IT186269 (n = 4.5), and a Chinese accession IT293155 (n = 4.5). By comparison, the average PPP and HSW were higher in Japanese adzuki beans, but the variation of each was not significantly different (at p < 0.05). In contrast, the average SPP was significantly lower in Japanese adzuki beans than in Korean and Chinese adzuki beans (p < 0.05). Based on their HSW, adzuki beans are classified as small (≤ 12 g), medium (12~18 g), and large (>18 g) seeds (Kim et al., 2011). Accordingly, most of the adzuki beans provided either small (f = 115) or medium (f = 122) size seeds. The remaining accessions (f = 15) provided large seeds. In many legumes, HSW is considered an important parameter to increase yield and is targeted during genetic improvement (Gerrano & Thungo, 2022). Therefore, the top five accessions including IT294637 (HSW: 21.47 g) from Japan, IT294311 (HSW: 20.37 g) and IT280298 (HSW: 19.60 g) from China, and IT236185 (HSW: 20.03 g) and IT186269 (HSW: 19.70 g) from Korea that yielded a greater HSW could be vital genetic materials. Among the control cultivars, Chungjupat yielded the largest PPP (n = 50.30) followed by Geomguseul (n = 39.30) and Arari (n = 24.30). In terms of SPP, Arari yielded the largest number while the other two produced comparable SPP (each 8.1). The HSW decreased in the order of Chunjupat (17.8 g) > Arari (15.9 g) > Geomguseul (11.90 g) (Table 2). Compared to the control cultivars, 21.43% of the accessions produced a higher PPP (n > 50.30), whereas only 3.97% of the accessions provided a higher SPP (n > 8.60). As stated before, PPP, SPP, and HSW are among the key determinant factors in terms of yield, and hence, adzuki bean accessions that simultaneously yielded a relatively higher PPP and SPP could be important genetic resources (Hu et al., 2022). In our study, ten accessions that yielded a relatively higher PPP (n ≥ 80.0) were identified. In particular, accessions IT216289, IT280307, IT216291, and IT280311 were selected as the best-performing accessions because they simultaneously produced a greater SPP (n ≥ 8.5) (Table 3).
Hierarchical cluster (HCA), Principal component (PCA), and correlation analyses
HCA and PCA are among the most popular multivariate statistical tools commonly used to differentiate huge populations of plant genotypes and determine the origins of variation. Furthermore, they are utilized to examine the relationship between variables (Rashid et al., 2020). In this study, HCA, PCA, and correlation analyses were conducted using the whole quantitative data set. As shown in Fig. 2A, the adzuki beans were clustered into three groups. Clusters I, II, and III contained 36, 145, and 71 accessions. Chinese accessions tend to dominate in cluster III (n = 45), whereas Korean accessions were abundant in cluster II (n = 68). Cluster I contained 8 accessions from China, 10 accessions from Japan, and 18 accessions from Korea (Supplementary Table S3). Among the variables, DF, D50F, and DM clustered together while SPP and PPP were also clustered together. The grouping of the adzuki bean accessions and the variables was further confirmed by PCA which yielded seven components that explained 100% of the variance (Fig. 2B, 2C). The first two principal components including component 1 (PC1) and component 2 (PC2) demonstrated 36.74 and 21.12% of the total variation, respectively. Hence, the distribution of the adzuki bean accessions was evaluated along the axes of PC1 and PC2 which yielded three regions of separation. The variance along PC1 was primarily based on DF (30.86%) and D50F (29.89%), while the variance along PC2 was related to HSW (36.81) and DM (31.72%). PPP contributed 11.39 and 3.31% for the variances along PC1 and PC2, respectively (Fig. 2D, Table 4). The loading plot of variables (Fig. 2C) was also in agreement with the HCA. As shown in Fig. 3 and Supplementary Table S3, statistical analysis demonstrated significant variations in all the quantitative variables except RG between the clusters (p < 0.05). The average DF and D50F decreased in the order of cluster I > II > III (p < 0.05). In contrast, the average DM was the longest in cluster II (110.32 days) followed by cluster I (107.19 days) and cluster III (102.44 days). On average, the highest PPP and SPP were observed in cluster I while the lowest were in cluster III. Likewise, the average HSW increased in the order of clusters I (7.38 g) < III (12.16 g) < II (13.98 g) (Fig. 3, Supplementary Table S3). In general, the HCA and PCA revealed the importance of quantitative agronomical traits in discriminating a large population of adzuki bean genetic materials (Ammar et al., 2015; Gerrano & Thungo, 2022).
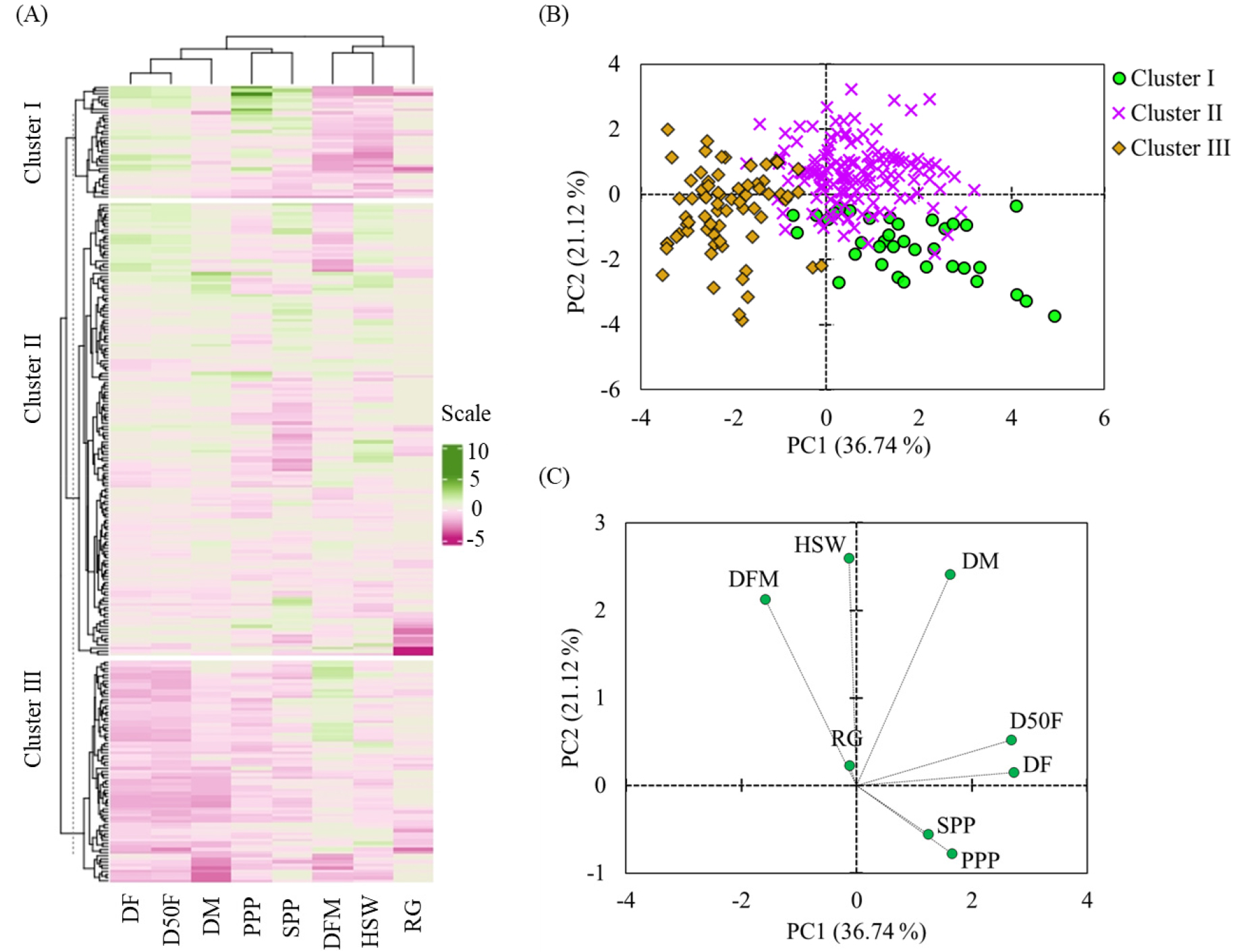
Fig. 2
Hierarchical cluster analysis (A), score plot of accessions (B), and loading plot of variables (C) from the principal component analysis. DF: Days to flowering, DFM: Days from flowering to maturity, DM: Days to maturity, D50F: Days to 50% flowering, RG: Rate of germination, HSW: One-hundred seeds weight, PPP: Pods per plant, SPP: Seeds per pod.
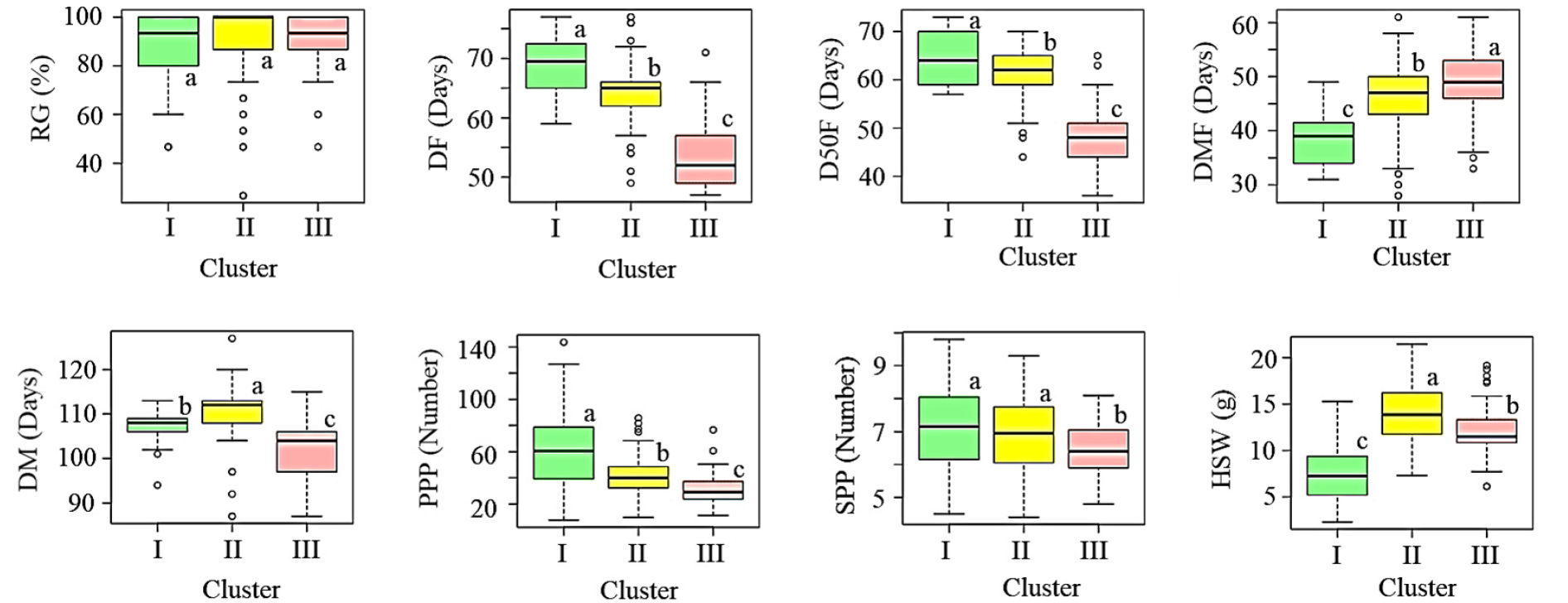
Fig. 3
Boxplots showing quantitative variations in agronomical traits between clusters. DF: Days to flowering, DFM: Days from flowering to maturity, DM: Days to maturity, D50F: Days to 50% flowering, RG: Rate of germination, HSW: One-hundred seeds weight, PPP: Pods per plant, SPP: Seeds per pod. Different letters on boxplots in a category indicate significantly different means (p < 0.05).
Table 4.
Individual and cumulative contributions of variables and the corresponding principal components when Eigenvalue is greater than one.
| Variables | Contribution of Principal components (%) | ||
| PC1 | PC2 | PC3 | |
| Rate of germination | 0.06 | 0.29 | 52.16 |
| Days to flowering | 30.86 | 0.12 | 2.38 |
| Days to 50% flowering | 29.89 | 1.48 | 2.21 |
| Days to maturity | 10.88 | 31.72 | 2.83 |
| Days from flowering to maturity | 10.44 | 24.58 | 11.34 |
| Pods per plant | 11.39 | 3.31 | 25.54 |
| Seeds per pod | 6.40 | 1.67 | 1.18 |
| One-hundred seeds weight | 0.07 | 36.81 | 2.37 |
| Eigenvalue | 2.94 | 1.69 | 1.17 |
| Variability (%) | 36.74 | 21.12 | 14.66 |
| Cumulative (%) | 36.74 | 57.86 | 72.52 |
Correlation analysis was also conducted based on origin and for the whole population and revealed some important associations between yield components and growth-related characters (Fig. 4). The germination rate had a negative or weak association with the rest of the variables regardless of origin. Interestingly, the growth-related variables including DF, D50F, and DM showed strong and positive correlations with each other (r ≥ 0.56, p < 0.05). Hu et al. (2022) also discovered positive relationships between growth-related variables in Korean adzuki beans, which coincided with our findings. In another investigation, Wang et al. (2001) reported negative or weak relationships in Chinese adzuki bean germplasms. Yield-related variables including PPP and SPP showed positive and significant associations with each other although the correlation coefficient values were far from +1 (0.26 ≤ r ≤ 0.31, p < 0.05). A recent study using cowpea genetic materials also found a positive link between yield-related components including SPP and PPP (Gerrano & Thungo, 2022). HSW showed a negative correlation with DF, PPP, and SPP, but had a positive and significant correlation with DM (r ≥ 0.27, p < 0.05), the findings being consistent with a recent report (Hu et al., 2022). Another interesting link was the positive and significant relationship between DM and PPP, indicating that a longer maturity time could result in a higher yield.
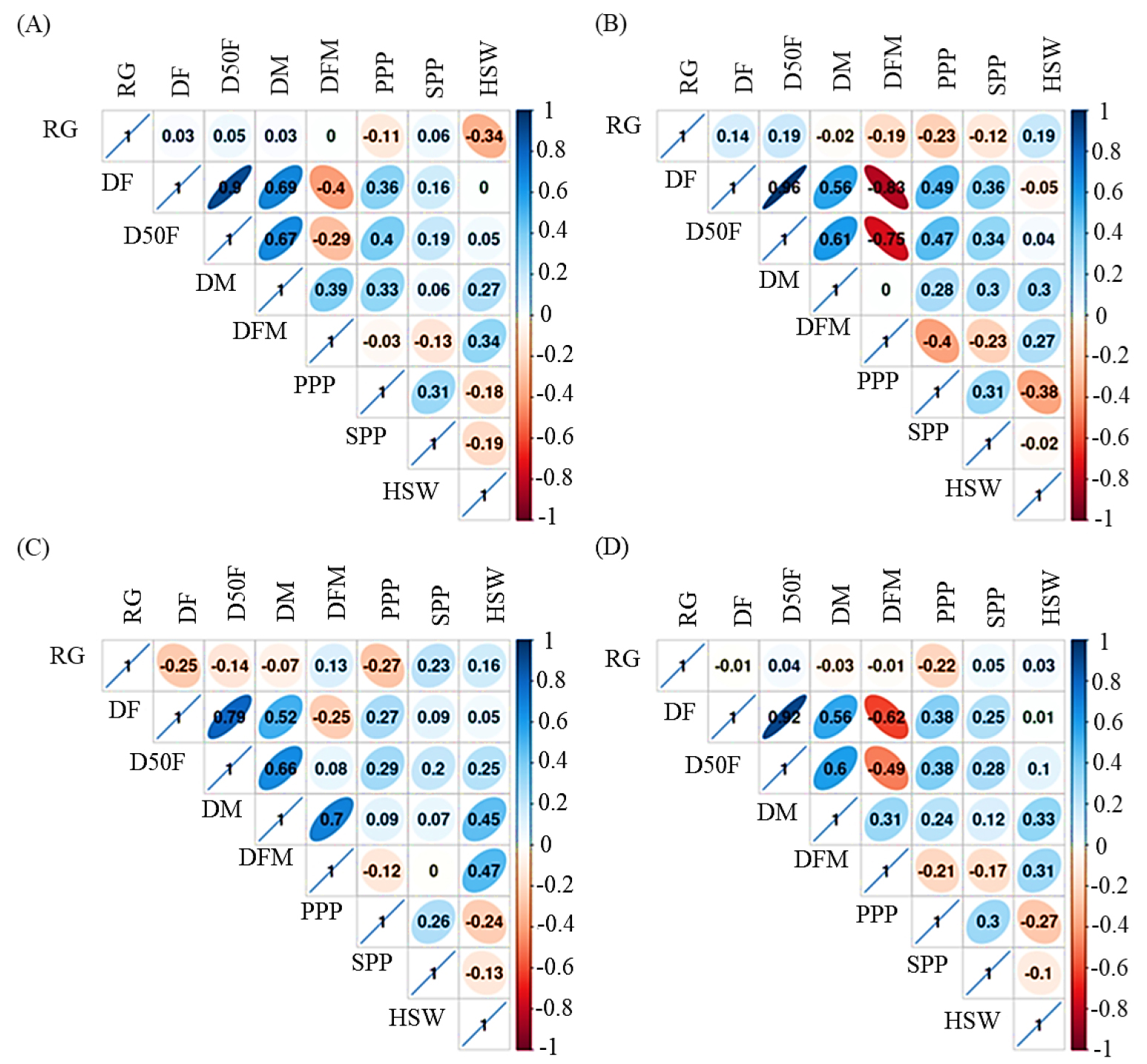
Fig. 4
Pearson correlation matrix of quantitative variables in Chinese (A), Japanese (B), Korean (C) adzuki beans, and the whole dataset (D). DF: Days to flowering, DFM: Days from flowering to maturity, DM: Days to maturity, D50F: Days to 50% flowering, GR: Rate of germination, HSW: One-hundred seeds weight, PPP: Pods per plant, SPP: Seeds per pod. Significant at p < 0.05 if r ≥ ±0.24.
To conclude, this study evaluated the genetic diversity of Chinese, Japanese, and Korean adzuki bean germplasms recently grown in Korea using agro-morphological traits. The adzuki beans showed diverse expressions of both qualitative and quantitative agronomical characteristics. Seed coat color, seed size, number of pods per plant, number of seeds per pod, and one-hundred seeds weight were among the most desirable traits that showed wide variations. Based on the average values, the top early maturing accessions and those yielding greater PPP and SPP were identified and hence, could be utilized for breeding and commercial use. The results of this study also signified the influence of origin on the agro-morphological properties of adzuki beans. The observed variations could initiate molecular-level investigations to view their genetic diversity in detail. Besides, exploring the differences in nutritional and non-nutritional metabolites according to the origin, seed size, and seed coat color would be of great interest which we are projected to conduct shortly.




