INTRODUCTION
MATERIALS AND METHODS
Plant material and growing conditions
Treatments
Experimental design
Morphological measurements
Physiology measurements
Biochemical measurements
Statistical analysis
RESULTS
Morphology of cucumber and cereals
Effects of four selected extracts on physiology of heat shock tolerance in cucumber seedlings
Effects of four selected extracts on biochemical of heat shock tolerance in cucumber seedlings
DISCUSSION
CONCLUSIONS
INTRODUCTION
Global warming is one of the hottest issues and has the potential to dramatically change environments worldwide (Vinagre et al., 2018). It is known that climate change is likely to impact species both directly and indirectly (Sun et al., 2018; Tong et al., 2017). Heat shock (HS) is defined as the exposure of plants to a sudden and significant temperature increase for a short period of time (Faralli et al., 2015). During the HS, plants are subjected to physical stress in their environment (Ruelland & Zachowski, 2010). The initial effects of high temperature on plants include growth reduction, water loss, a change in photosynthetic efficiency, and oxidative stress (Hasanuzzaman et al., 2013). Longer periods of high temperature stress may result in wilting, necrosis, leaf pigmentation loss for herbaceous plants, as well as leaf elongation repression (Scafaro et al., 2010). Therefore, heat stress is considered as a problem for agriculture throughout the world (Carmo-Silva & Salvucci, 2012; Jha et al., 2014), and it may lead to dramatic losses in crop yields (Lesk et al., 2016).
Cucumber (Cucumis sativus L.), one of the most important vegetables, was widely cultivated in different latitudes of the world. The optimal growth temperature for cucumber is 25-28°C (Liu et al., 2021). Long-term high temperatures (HT) above 35°C significantly inhibit the growth and development of cucumber seedlings, ultimately leading to a decrease in the quality and commercial value of cucumber fruits (Dong et al., 2020; Yu et al., 2018; Yu et al., 2022). When ambient temperatures reach 50°C, leaf and stem undergo rapid wilting, root growth is retarded, pollen viability is diminished, the fruit is deformed, and it might even result in plant death, all leading to compromised fruit quality and commodity yield (Yan et al., 2022). Among the main abiotic stresses, HT limits the yield and quality and yield of agricultural plants, including cucumber (Gong et al., 2020; Saini et al., 2022). In previous studies, many physiological indexes, including the content of superoxide dismutase (Liu et al., 2021), the electrical conductivity (Yu et al., 2022), and the photosynthetic efficiency (Xu et al., 2018), were used to test the heat tolerance performance of cucumber at the seedling stage.
In recent years, the demand from consumers for organic foods has increased. Therefore, scientists must try to find alternatives to these synthetic chemicals to ensure the production of safer and more environmentally friendly horticultural production. These plant extracts include diverse formulations of compounds and substances that are applied to seeds, plants or soils to improve crop vigor, yields, quality, and tolerance to abiotic stresses. Additionally, plant extracts are gaining widespread attention and are increasingly integrated into high-value production systems (e.g., greenhouse production, fruit, vegetable, and floriculture), thereby sustainably increasing productivity and quality (Zulfiqar et al., 2020).
Additionally, the use of allelopathic plant water extracts also provides a bio-safe and eco-sustainable alternative for crop development under stress (Bajwa et al., 2018). The effect of the application of allelochemicals, either for growth stimulation or retardation, is dose-dependent. Hormesis is a biphasic dose- response relationship with low doses including stimulatory effects by activating adaptive mechanisms that enhance resilience, while high doses may induce inhibitory responses that at even higher doses often become toxic (Trejo-Téllez et al., 2020). In other studies, aqueous garlic extract has been shown to alter the antioxidative response of cucumber seedlings, and this effect was observed to be concentration-dependent. The other reported that garlic (shoots, roots and bulbs) exhibits a “hormesis effect” at increased concentrations (Cheng et al., 2011; Wang et al., 2009). The effects of various leaf extracts (garlic and green tea, each at two different concentrations) on the growth and seed yield of guar (Cyamopsis tetragonoloba) showed positive effects when applying green tea extracts at 150 ppm and garlic extract at 300 ppm (Abdou & Bakhtiarizade, 2019). Garlic root exudates at low concentrations (0.1 and 0.2 g/mL) have been demonstrated to promote lettuce seed germination and seedling growth, but high concentrations (0.4 and 0.6 g/mL) have shown inhibitory effects (Zhou et al., 2011).
During the past few years, it has been well established that plant extracts improve seedling growth and physiological and biochemical traits; the positive influence was highly dose- dependent; however, there is a lack of studies on the guava, garlic, aloe, and alfalfa extracts under heat shock conditions. Therefore, the present study was conducted to select a heat- sensitive test crop of cereals and vegetable and to evaluate the effective concentration of four selected extracts for hormetic promotion based on the morphological responses to heat stress in a preliminary trial. In addition, the physiological and biochemical mechanisms underlying the heat tolerance of cucumber under the influence of selected extracts were to be optimized.
MATERIALS AND METHODS
Plant material and growing conditions
Cucumber (cv. Ho Dong Cheong Jang F1), barley (cv. Saechalssalbori), and maize (cv. Mibaek 2 Ho) were purchased from Asia Seed Co., Ltd in South Korea. Rice seeds (cv. Hopyeong) were provided by the Jeollanamdo Agricultural Research and Extension Service. For cucumber, maize, and barley crops, a commercial horticultural soil (No. 2 Sunghwa, Boseong, South Korea) consisting of zeolite (5%), perlite (8%), peat moss (8%), vermiculite (12%), cocopeat (67%), and fertilizer (0.4%) was used. For rice, a commercial soil (No. 1 SungHwa, Boseong, South Korea) consisting of zeolite (8%), diatomaceous earth (8%), vermiculite (30%), decomposed granite (21.6%), cocopeat (32%) and fertilizer (0.38%) was used.
The maize and cucumber seeds were sown into 50-cell plastic trays, and the rice and barley seeds were sown into plain plastic trays with different commercial soil. At the two-three-leaf stage, maize and cucumber crops were transplanted into plastic pots (16 × 6 cm) with 200 g of commercial soil. At the three-four-leaf stage, barley and rice crops were transplanted into plastic pots (6 × 6 cm) with 130 g of commercial soil. After that, these transplanted plants were maintained under glasshouse with natural daylight. The greenhouse conditions consisted of 14 h of light and 10 h of darkness, with a day/night temperature range of 30 ± 2°C/ 20 ± 3°C, respectively, 70% relative humidity, and photosynthetically active radiation (PAR) of 500 µmol m-2 s-1.
Treatments
According to the previous studies (Ei et al., 2024; Ei et al., 2025), guava, aloe, garlic and alfalfa extracts were the most effective in promoting the growth of rice and vegetables. Therefore, these four extracts were selected to confirm that the effect of these extracts is also promoted under HS conditions. The study was based on four water extracts: guava (Psidium guajava L.), aloe (Aloe vera (L.) Burm. f.), garlic (Allium sativum L.) and alfalfa (Medicago sativa L.) extracts according to the protocol described in a previous study (Ei et al., 2024). At the two- to three-leaf stage, these extract treatments were applied using the soil application method with concentrations of 0.05, 0.1, 0.5, and 1%. For each plant sample, 10 mL of the extract was applied. Distilled water was taken as control treatment.
The next day, all pots were placed in a growth chamber (HB-303DH-O, HANBAEK SCIENTIFIC, Bucheon, Korea) to start the HS treatment. This growth chamber was set up at 16 h of light and 8 h of darkness with 35°C/25°C day/night temperature, and exposed to light with a photon flux density of photosynthetically active radiation (PPFD) of 575 µmol m-2 s-1 for 5 days. To avoid water stress in plants, water was given twice a day. The control consisted of normal-, and heat conditions in the growth chamber. After the 5-day HS treatment, the pots were returned to the growth room under normal conditions (28/18°C for 14/10 h) (Tripathee, 2008).
Experimental design
In a preliminary trial, the study was based on two factors: various concentrations of guava, aloe, garlic and alfalfa extracts (0.05, 0.1, 0.5 and 1%) and four different crops, cucumber, maize, barley and rice crops. Each treatment had three replicates and replicates contained 6 plants in maize and cucumber crops, 9 plants in barley and rice crops. In this study, treatments were arranged factorial design with two factors and each treatment was repeated two times.
In physiology and biochemical tests, the experiment was designed on one factor: 0.1% concentration of four selected extracts treated on cucumber in heat conditions. Treatments were arranged in a completely randomized design with one factor and each treatment was repeated 2 times.
Morphological measurements
In this study, cucumber (Cucumis sativus L.), maize (Zea mays L.), barley (Hordeum vulgare L.) and rice (Oryza sativa L.) crops were used to select the HS sensitive crop. On the 5 day after HS treatment, the plant height and the fresh weight of the shoots were measured. The visual assessment of plant damage was carried out daily following the 1-5 scale (Table 1).
Table 1.
Visual assessment (1-5 scale) based on injury percentage.
Physiology measurements
The cucumber variety was used as a test plant to evaluate recovery under heat stress and it showed the highest recovery rate when treated with the selected extracts. Our previous studies (Ei et al., 2024) show that four water extracts (P. guajava, A. vera, A. sativum and M. sativa) at a concentration as low as 0.1% were evaluated for their efficacy in reducing heat stress in cucumber plants. The other procedures were the same as those described in previous findings at 3 and 5 days after treatment (DAT) under HS conditions.
RWC was measured in the broad second leaves of cucumber seedlings on the 3 and 5 DAT under HS conditions. Each paper bag was weighed (W1) before the leaf samples were added. The leaf samples were immediately cut and placed in each paper bag and then weighed together with the paper and fresh leaf samples (W2). These leaf samples were carefully dried in paper bags at 70°C for 24 hours, then the samples were weighed together with the paper bags and dry leaf samples (W3). The value of RWC was calculated according to the formula [(W2 – W3) / (W2 – W1)] × 100 (Rai et al., 2016).
The chlorophyll fluorescence parameter (Fv/Fm), which indicates the maximum quantum efficiency of photosystem II (PSII), and the total chlorophyll and carotenoid contents of cucumber were evaluated three and five days after treatment under HS conditions. The chlorophyll fluorescence of PSII was measured using a portable pulse modulation fluorometer (PAM-2500, Heinz Walz GmbH, Effeltrich, Germany). Prior to the measurements, the leaves were adapted to darkness for 15 mins to ensure that all antenna pigments were fully open. The quantification of chlorophyll and carotenoid content was performed spectrophotometrically at 665, 652 and 470 nm according to the methodology described in Ei et al. (2024).
Biochemical measurements
Determination of oxidative damage
The H2O2 concentration was determined according to the methodology described by Jana & Choudhuri (1981). Cucumber leaves (0.5 g) were first pulverized in liquid nitrogen and then homogenized in 3.75 mL of phosphate buffer at a concentration of 50 mM (pH 6.8). The homogenate was centrifuged at 6,000 × g for 25 min. For the determination of H2O2, 1 mL of 0.1% titanium sulfate in 20% (v/v) H2SO4 was added to the supernatant and centrifuged at 6,000 × g for 15 min. The absorbance of the supernatant at 410 nm was recorded and the absorbance values were calibrated using a standard curve generated with known H2O2 concentrations. The H2O2 concentration was determined using the extinction coefficient ε = 0.28 µmol-1 cm-1.
The MDA levels were determined using the thiobarbituric acid (TBA) method according to Kuk et al. (2002). Cucumber leaves (0.5 g) were finely ground and mixed with 25 mL of 0.5% TBA in a 20% trichloroacetic acid (TCA) solution. The solutions were then centrifuged at 20,000 × g for 15 min, after which the supernatant was collected. The mixture was then heated in boiling water to 100°C for 25 min and then immediately cooled to stop the reaction. After cooling, the mixture was centrifuged at 20,000 × g for 15 min. The absorbance values of the supernatant at 440 nm, 532 nm and 660 nm were determined using a UV spectrometer (SPECTROstar Nan, BMG LABTECH, GmbH, Offenburg, Germany). MDA levels were calculated using an extinction coefficient ε = 156 mm-1 cm-1 and the following formula (Du & Bramlage, 1992):
{[(A532-A600)-[(A440-A600) (MA of sucrose at 532 nm/MA of sucrose at 440 nm)]]/157000}106
Determination of antioxidant activities
A 100 mM buffer solution (pH 7.5) containing 2 mM EDTA, 1% PVP and 1 mM phenylmethylsulfonyl fluoride (PMSF, C7H7FO2S) was added to 3.75 mL of approximately 0.5 g of frozen leaves to extract the enzymes from them. The mixture was then crushed with a pestle and mortar before being placed in a refrigerated centrifuge (VS-24SMTI, high-speed refrigerated centrifuge, Vision Scientific Co., Ltd., Daejeon, South Korea) at 14,000 × g for 20 min.
SOD activity was measured according to a method (Giannopolitis & Ries, 1977), with modifications as described in (Chowdhury & Choudhuri, 1985; Zhang et al., 1995). A total volume of 300 µL was obtained by adding 20 µL of the enzyme extract to a reaction mixture containing 230 µL of nitrogen blue tetrazolium (NBT), 260 mM riboflavin, 260 µM methionine, 2 mM ethylenediamine tetraacetic acid (EDTA) and 200 mM phosphate buffer at pH 7.0. After 10 min of exposure at an intensity of 78 mmol photon s-1 m-2, the absorbance at 560 nm of the mixture in the test tubes was measured. The concentration of SOD enzyme required to suppress the degradation rate of NBT by 50 at 560 nm is referred to as one unit of SOD activity (SPECTROstar Nano).
The CAT and GPOD were determined using previously published methods (Chance & Maehly, 1995; Fu & Huang, 2001). Spectrometer measurements were performed using the SPECTROstar Nano at 240 nm for a duration of 1 min. These readings were used to evaluate the CAT activity quantifying the rate of H2O2 degradation. The reaction mixture contained 150 µL of 200 mM phosphate buffer (pH 7.0), 15 µL of 15 mM H2O2, 120 µL of distilled water and 15 µL of enzyme extract, giving a total volume of 300 µL with which to start the reaction. The oxidation of guaiacol during GPOD activity was assessed by monitoring the increase in absorbance at 470 nm over a period of 1 min. The reaction mixture consisted of 5 µL 200 mM guaiacol, 280 µL 200 mM phosphate buffer (pH 7.0) and 15 µL 40 mM H2O2.
The determination of AsA was carried out according to the methods (Law et al., 1983). Plant samples (0.5 g) were mixed with 5 mL of 5% metaphosphoric acid and then centrifuged at 13,000 × g for 15 min at 4°C. To prepare total ascorbic acid (AsA), combine 100 µL of the sample with 250 µL of a solution containing 5 mM EDTA and 150 mM potassium phosphate buffer (pH 7.4) and add 50 µL of 10 mM DTT (dithiothreitol). To another AsA, add 100 µL of the sample, 250 µL containing 5 mM EDTA and 150 mM potassium phosphate buffer (pH 7.4) and 50 µL of distilled water instead of DTT. After thorough mixing and a 10 min incubation at room temperature, 50 µL of 0.5% NEM (N-ethylmaleimide) was added. Both samples were vortex-mixed and incubated for 60 s at room temperature. Each sample was supplemented with 611 µL consisting of 200 µL of 10% TCA (trichloroacetic acid), 200 µL of 44% phosphoric acid, 200 µL of 70% ethanol combined with 4% bipyridine and 11 µL of 30% FeCl3. After vortex mixing, the samples were incubated at 37°C for 60 min, and the absorbance was recorded at 525 nm. A standard curve from 0 to 100 µM L AsA was used for calibration.
The measurement of GSH was performed using an enzymatic cycling assay method as described in Gaitonde, 1967, with some modifications. The plant samples (0.5 g) were extracted with 6 mL of 5% sulfosalicylic acid and then centrifuged at 12,000 rpm for 10 min at 4°C. For total glutathione analysis, 20 µL of the supernatant was combined with 170 µL of distilled water and 700 µL of daily buffer consisting of a 100 mM sodium phosphate buffer containing 6.3 mM EDTA (pH 7.5) and 0.248 mg/mL NADPH (nicotinamide adenine dinucleotide phosphate). Subsequently, 6 mM DTNB (5,5’-dithio-bis-(2-nitrobenzoic acid) was prepared in the daily buffer and 100 µL was added. The reaction started with the addition of 10 µL of 0.5 units of glutathione reductase and the increase in absorbance at 412 nm was recorded at 1 min.
Statistical analysis
The experiment was set up in a factorial layout with three replications in a completely randomized design. Multiple comparisons were performed to assess differences in variables between components. Significant differences were assessed using an analysis of variance (ANOVA) in a statistical software program (Statistix version 8.0). When a significant difference was found, the means were distinguished using Tukey’s honestly significant difference (HSD) and least significant difference (LSD) tests at α = 0.05.
RESULTS
Morphology of cucumber and cereals
After three days of heat stress, all plants tested showed signs of stress, including brown edges, wilting and stunted growth (data not shown). In contrast, the control plants showed robust growth and their green foliage testified to their vigor. All plants exposed to the stress showed drooping leaves, but the control plants remained upright and spread their leaves. The plants exposed to heat stress showed a greater divergence between their petioles and leaves than the control plants, which remained upright and stable. The leaf veins of the stressed plants were greener in color compared to those of the control plants.
All of the extract treatments resulted in significant differences in shoot fresh weight and plant height of cucumber, but there were no significant differences in maize, barley, and rice (Tables 2, 3, 4, 5). The injury to the studied plants increased by 50%, 40%, 30%, and 50% in stress treatment alone, but it raised by 20-30%, 10-20%, 20-30%, and 15-30% in extract treatments for cucumber, maize, barley, and rice compared to the control group. All extract treatments decreased the HS injury rate of cucumber 20-30%, maize 20-30%, 0-15%, and 20-25% compared to HS alone.
Table 2.
Effects of selected plant-derived biostimulants on the percentage of injury, plant height, and shoot fresh weight of cucumber under heat shock (HS) condition.
Means within a column followed by the same letters are not significantly different at the 5% level according to Tukey’s HSD test. DAT; days after treatment. Pg; P. gaujava extract. Av; A. vera extract. As; A. sativum extract. Ms; M. sativa extract. Treat; treatment. Conc; concentration. ***; significant at 0.001 level. *; significant at 0.05 level. ns; non-significant. Pr; probability value, F; F-statistic from ANOVA, CV; Coefficient of variation. Parentheses indicate the percentage of the control.
Table 3.
Effects of selected plant-derived biostimulants on the percentage of injury, plant height, and shoot fresh weight of maize under heat shock (HS) condition.
Means within a column followed by the same letters are not significantly different at the 5% level according to Tukey’s HSD test. DAT; days after treatment. Pg; P. gaujava extract. Av; A. vera extract. As; A. sativum extract. Ms; M. sativa extract. Treat; treatment. Conc; concentration. ***; significant at 0.001 level. *; significant at 0.05 level. ns; non-significant. Pr; probability value, F; F-statistic from ANOVA, CV; Coefficient of variation. Parentheses indicate the percentage of the control.
Table 4.
Effects of selected plant-derived biostimulants on the percentage of injury, plant height, and shoot fresh weight of barley under heat shock (HS) condition.
Means within a column followed by the same letters are not significantly different at the 5% level according to Tukey’s HSD test. DAT; days after treatment. Pg; P. gaujava extract. Av; A. vera extract. As; A. sativum extract. Ms; M. sativa extract. Treat; treatment. Conc; concentration. ***; significant at 0.001 level. **; significant at 0.01 level. ns; non-significant. Pr; probability value, F; F-statistic from ANOVA, CV; Coefficient of variation. Parentheses indicate the percentage of the control.
Table 5.
Effects of selected plant-derived biostimulants on the percentage of injury, plant height, and shoot fresh weight of rice under heat shock (HS) condition.
*Means within a column followed by the same letters are not significantly different at the 5% level according to Tukey’s HSD test. DAT; days after treatment. Pg; P. gaujava extract. Av; A. vera extract. As; A. sativum extract. Ms; M. sativa extract. Treat; treatment. Conc; concentration. ***; significant at 0.001 level. ns; non-significant. Pr; probability value, F; F-statistic from ANOVA, CV; Coefficient of variation. Parentheses indicate the percentage of the control.
Consequently, stress-only treatment diminished shoot fresh weight by 58%, 58%, 57%, and 36%, and plant height by 33%, 30%, 32%, and 22% in cucumber, maize, barley, and rice, respectively, in comparison to control plants. When treated with extract, shoot fresh weight reduced by 17-31%, 34-50%, 31-43%, and 14-35%, while plant height decreased by 7-21%, 8-22%, 7-31%, and 7-21% in cucumber, maize, barley, and rice, respectively, compared to control plants. However, compared to stress-only treatment, the extract application increased shoot fresh weight by 27-41%, 8-24%, 14-26%, and 1-22% and increased plant height by 12-26%, 8-22%, 1-25%, 1-15% in cucumber, maize, barley, and rice, respectively. The growth parameters observed for the plant extracts in combination with the heat treatment were significantly better than those resulting from the heat treatment alone, suggesting that 0.1% P. guajava extracts confer a protective benefit to plants under heat stress conditions.
The ANOVA showed a significant effect of extract concentration (p < 0.001), although treatment and the interaction between treatment and concentration were not significant (p > 0.05). Tukey’s HSD test indicated significant differences between certain concentration levels; however, these differences did not show a consistent concentration-dependent pattern across all parameters. This suggests that although concentrations influence the measured parameters, plant responses may exhibit threshold or saturation effects rather than a simple linear correlation. The lack of a clear trend in the dose-response relationship may be due to biological variability or threshold effects in plant responses, where increasing the concentration beyond a certain level does not cause further significant changes. Therefore, although the concentration influences the response, the correlation is not completely linear.
Effects of four selected extracts on physiology of heat shock tolerance in cucumber seedlings
The relative water content in the leaves was an indicator of the moisture content in the leaves and was directly influenced by the water content in the soil. The application of stress led to a reduction in the relative water content (RWC) in the leaves of cucumber seedlings exposed to HS; however, the effects were more pronounced in the single stress treatment (Fig. 1A). When the control treatment exhibited 98% RWC, the stress-only treatment demonstrated 85% RWC, indicating a reduction of 13% in RWC. Furthermore, the extract treatment plants exhibit an RWC of 90-95%, which was diminished by 3-8%. Consequently, the extract treatment recovered the RWC rate by at least 5-10% under heat conditions in comparison to the stress-only treatment. The plants treated with P. guajava and A. sativa extracts had the highest RWC under HS compared to the stress treatment alone.

Fig. 1.
Effects of selected plant-derived biostimulants on relative water contents (A), chlorophyll fluorescence, Fv/Fm (B), total chlorophyll content (C) and total carotenoids content (D) of cucumber under heat shock (HS) at 3 and 5 days after treatment (DAT). Pg, P. guajava; Av, A. vera; As, A. sativum; Ms, M. sativa extracts. Each bar represents the mean value. Bars with the same letters are not significantly different at the 5% level according to the Least Significant Difference test.
The effect of plant extracts on HS cucumbers was investigated by measuring photosynthesis-related parameters, including chlorophyll fluorescence (Fv/Fm) and pigment content (total chlorophyll and carotenoid contents) (Fig. 1B). The Fv/Fm values remained largely unchanged under HS across treatments, although cucumber plants treated with P. guajava extract had significantly higher Fv/Fm at 3 DAT, suggesting enhanced PSII functionality. No significant differences were observed among the treatments by 5 DAT. On 3 DAT, no significant differences in total chlorophyll content were observed among the extract-treated plants and the HS control, suggesting that HS alone may have contributed to a temporary increase in chlorophyll content. However, on 5 DAT, all plant extract treatments resulted in a significant increase in total chlorophyll content compared to the stress-only plants. Among the treatments, P. guajava extract had the highest chlorophyll levels under HS (Fig. 1C). In contrast, the total carotenoid content varied significantly with the application of the extract. Notably, A. vera extract had the highest carotenoid contents on both 3 and 5 DAT under HS (Fig. 1D).
Effects of four selected extracts on biochemical of heat shock tolerance in cucumber seedlings
The cucumber plants showed a strong increase in H2O2 concentration and MDA levels when grown with or without the extracts during HS (Fig. 2). The stress treatment alone significantly increased the H2O2 content in cucumber under HS conditions compared to the control, followed by applied A. vera and M. sativa extract, resulting in increased oxidative damage by relative oxygen species (ROS). However, the application of P. guajava and A. sativum extracts to the soil significantly reduced H2O2 content under HS conditions compared to the control. The stress treatment alone had the highest MDA levels in cucumber under HS conditions compared to the control, followed by A. vera and P. guajava extracts. On the other hand, A. sativum and M. sativa extracts alleviated oxidative stress and consequently reduced MDA levels under HS conditions.
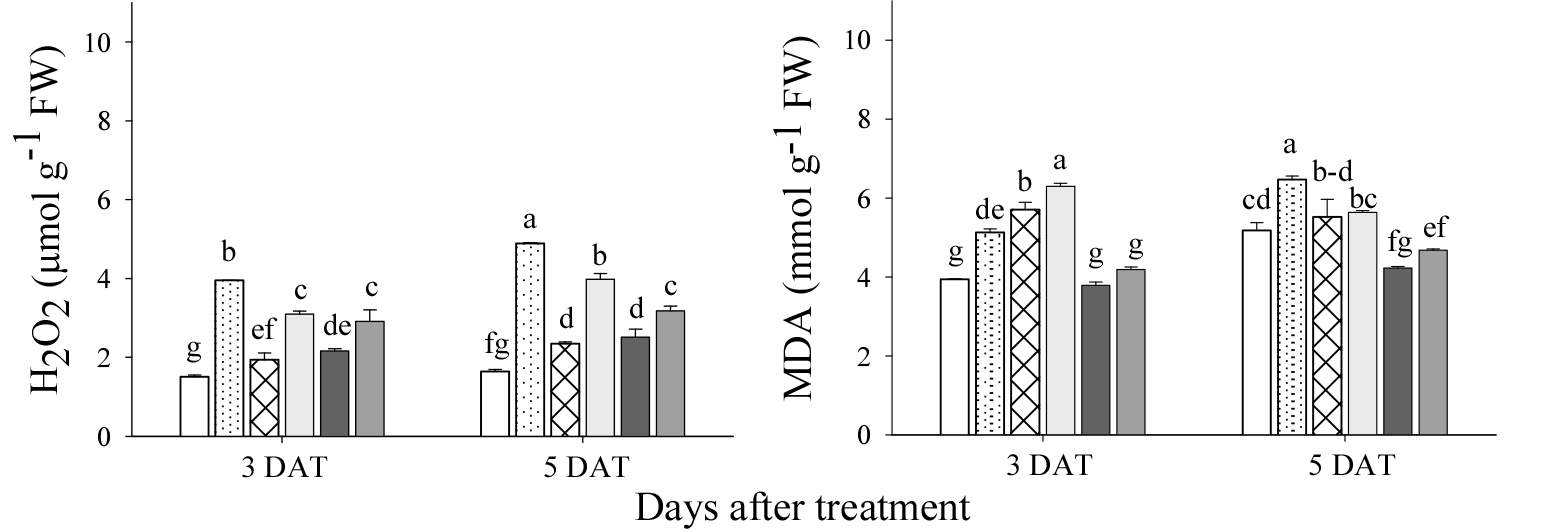
Fig. 2.
Effects of selected plant-derived biostimulants on H2O2 and MDA of cucumber under heat shock (HS) at 3 and 5 days after treatment (DAT). Pg, P. guajava; Av, A. vera; As, A. sativum; Ms, M. sativa extracts. Each bar represents the mean value. Bars with the same letters are not significantly different at the 5% level according to the Least Significant Difference test.
The results showed that SOD, CAT, GPOD activity, and AsA and GSH contents were upregulated under HS conditions when treated with any of the extracts as opposed to only stress or control (Fig. 3). The most increased SOD activity was found in cucumber plants treated with extracts of P. guajava and A. sativum, followed by those treated with extracts of A. vera and M. sativa, compared to the control group. The CAT activity observed in the leaves of cucumber seedlings was highest under HS treatment, A. vera and M. sativa extract, while it was lowest under HS in P. guajava and A. sativum extract compared to the control group. The activity of the enzymes GPOD, AsA and GSH showed a greater increase in all extract treatments than in the plants exposed to HS alone and in the control group. The cucumber seedlings treated with extracts of P. guajava and A. sativum showed the highest levels of GPOD, AsA and GSH. This was followed by the extracts of A. vera and M. sativa on both days of HS evaluation compared to the control group.
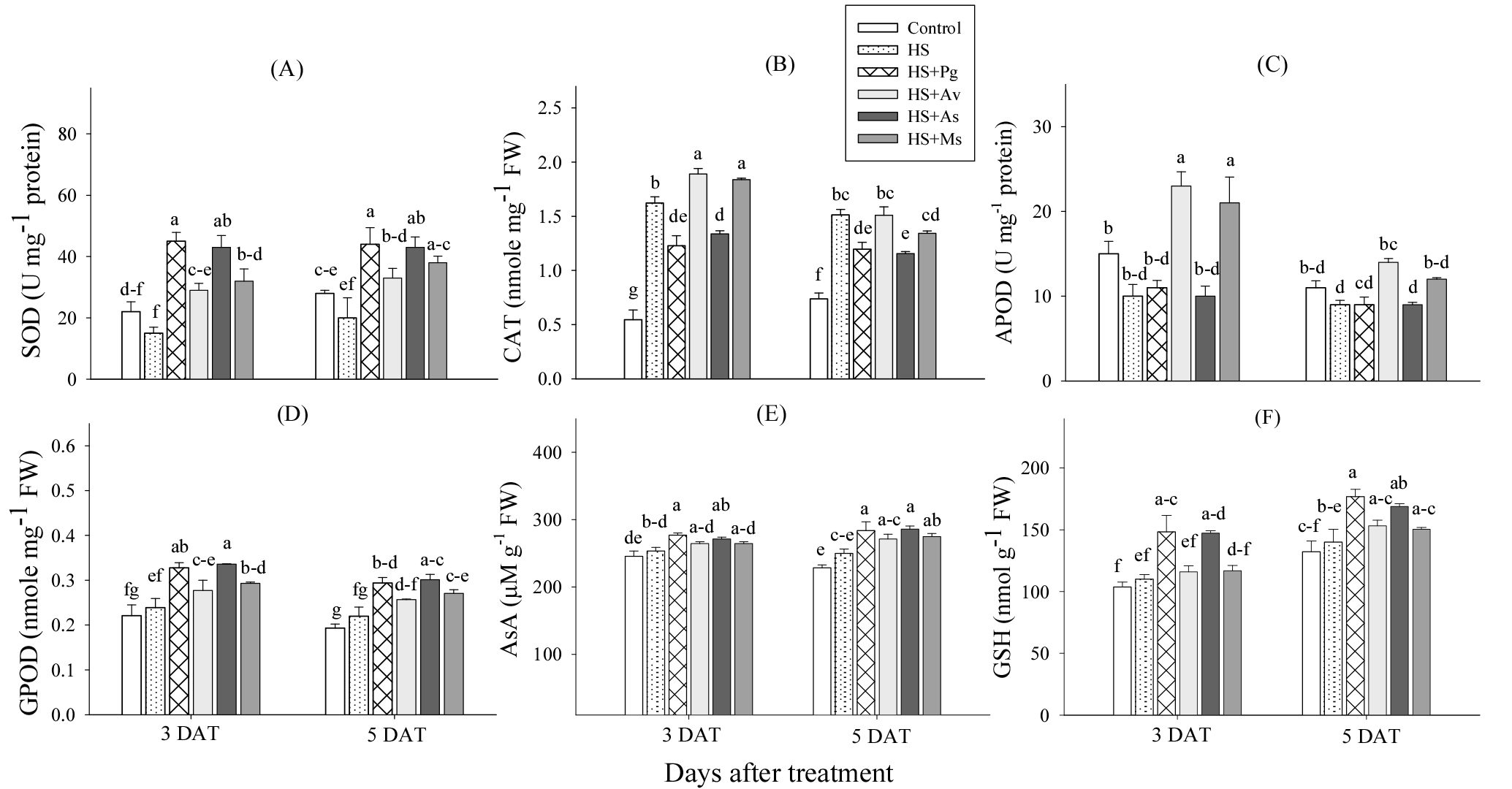
Fig. 3.
Effects of selected plant-derived biostimulants on the SOD (A), CAT (B), GPOD (C), AsA (D), and GSH (E) contents of cucumber under heat shock (HS) at 3 and 5 days after treatment (DAT). Pg, P. guajava; Av, A. vera; As, A. sativum; Ms, M. sativa extracts. Each bar represents the mean value. Bars with same letters are not significantly different at the 5% level according to the Least Significant Difference test.
DISCUSSION
Heat stress is a major abiotic stressor that affects plant growth and productivity and leads to numerous physiological and biochemical changes (Bita & Gerats, 2013). HS causes overgrowth, leaf dryness and fruit malformation, which negatively impact cucumber quality and yield (Wang et al., 2023). For cereal plants, the damage caused by the stress alone increased by 30-50%, and by the application of the extract by 0-30%. Plant height decreased by 22-32% and shoot fresh weight decreased by 36-58% in the stress-only treatment compared to the control. However, the extract treatments increased plant height by 1-25% and shoot fresh weight by 1-26% compared to the stress-only treatment (Tables 2, 3, 4, 5). The fact that cereals showed a comparatively lower response suggests that monocotyledons may rely on other physiological pathways to adapt to stress, making them less susceptible to plant extracts (Wang et al., 2022).
In the cucumber plants studied, damage injury increased by 50% under the stress conditions alone and by 30-50% when extracts were applied, so that the application of extracts reduced the damage injury of HS. The plant height of the cucumber decreased by 33% and the shoot fresh weight decreased by 58% in the stress treatment alone compared to the control. Conversely, plant height of the cucumber increased by 12-26% and shoot fresh weight increased by 27-41% in the extract treatment compared to the stress-only treatment (Tables 2, 3, 4, 5). Previous studies have reported similar results, suggesting that plant biostimulants are more effective in vegetable crops than in cereals. This difference can be attributed to differences in root architecture and nutrient uptake mechanisms (Ertani et al., 2017; González-González et al., 2020). The significant effect on cucumber suggests that broad-leaved vegetable plants may be more responsive to treatment with plant extracts compared to cereals, possibly due to differences in metabolic pathways, cuticle permeability and root absorption efficiency (Bulgari et al., 2019; Colla et al., 2015).
The findings of this study show that extracts of P. guajava, A. vera, A. sativum and M. sativa significantly improve the growth parameters of cucumber and cereals under heat stress conditions (Tables 2, 3, 4, 5). Previous research (Calvo et al., 2014; Yakhin et al., 2017), plant extracts containing bioactive components can improve plant growth and reduce the effects of abiotic stress. The compounds in question likely improve stress tolerance through antioxidant activity, osmoprotection and modulation of phytohormone signaling (Ertani et al., 2018; Khan et al., 2020). Application of extracts of P. guajava and A. sativum to cucumber plants significantly improved their ability to recover from HS, indicating a biostimulant effect.
Previous studies have shown that these extracts were most beneficial when applied to the soil in rice and vegetable crops (Ei et al., 2024; Kunicki et al., 2010). This study shows that all concentrations alleviate HS by increasing plant height and fresh shoot weight in the tested plants across all extracts. All extracts showed a positive effect on the tested plants at a concentration of 0.1% compared to a pure stress treatment. Environmental factors, dosage amount, and timing of application were also found to influence the effectiveness of extract applications (Yildirim et al., 2008).
The decrease in RWC under HS shows remarkable deterioration in the water status of cucumber seedlings, with a 15% decrease observed in the treatment with only one stress factor compared to the control group (Fig. 1A). The cucumber plants treated with water extracts of P. guajava and A. sativum showed an increase of 8% in RWC when exposed to heat stress alone, emphasizing their function in reducing water loss at elevated temperatures. This effect can be attributed to the abundant A. vera extract, which could help improve RWC in cucumber under HS (Mohamed et al., 2022). The other biostimulants, including olive leaf extract, have shown the ability to increase RWC and stress tolerance in cucumber under heat stress conditions (Dai et al., 2012). This is in line with previous studies on cucumber, which indicate that HS leads to a reduction in relative water content due to increased transpiration and impaired water uptake (Camele et al., 2017; Hongal et al., 2023).
In this study, plant extracts were shown to be able to reduce HS in cucumbers by increasing the chlorophyll fluorescence Fv/Fm, the total chlorophyll and carotenoid contents (Fig. 1B, C, D). P. guajava extract showed an increased chlorophyll fluorescence Fv/Fm at 3 DAT, and the total chlorophyll contents at 5 DAT when exposed to HS alone. This is in line with the findings that guava leaf extracts enhance photosynthetic activity in plants subjected to HS (Zhang et al., 2019). The results show that A. vera extract produced the highest total carotenoids under HS conditions on both days of measurement (Fig. 1D). The results are consistent who found that A. vera and other plant extracts can act as natural antioxidants to protect photosynthetic pigments and make plants more resistant to stress (Noreen et al., 2020). Our results are also consistent in that biostimulants from plant extracts improve physiological parameters such as the concentration of carotenoids and chlorophyll during HS, which supports the plant's ability to recover (Devireddy et al., 2023).
This study shows that stress-only treatment showed increased levels of H2O2 and MDA, confirming the negative effects of HS (Fig. 2). The increased H2O2 concentrations in the extracts of A. vera and M. sativa and the increased MDA concentrations in the extracts of A. vera and P. guajava indicate a possible oxidative stress response induced by their metabolites, possibly due to insufficient antioxidant capacity to mitigate ROS under HS conditions. Conversely, the extracts of P. guajava and A. sativum showed a significant decrease in H2O2 concentration, while the extracts of A. sativum and M. sativa reduced MDA concentration, emphasizing their strong antioxidant properties. The foliar application of trehalose (5-aminolevulinic acid) has been shown to improve antioxidant enzyme activities and lower ROS levels in tomatoes exposed to HS (Hasanuzzaman et al., 2019), confirming the results of this study. Heat stress leads to a significant increase in ROS, such as H2O2, which plays a role in oxidative damage and lipid peroxidation of membranes, as evidenced by increased MDA levels (Repek et al., 2022). The results show that the effects on the regulation of oxidative stress vary depending on the species and extract.
The results indicate that plant extracts significantly improved the antioxidant activity in cucumber plants during HS (Fig. 3). The extracts of P. guajava and A. sativum showed the highest activities of SOD, GPOD, AsA and GSH, indicating a strong response to oxidative stress. However, the extracts of A. vera and M. sativa significantly increased CAT and GPOD activity in cucumber plants exposed to HS. Other findings discovered that A. nodosum extract increased SOD and CAT activity in soybean plants exposed to HS, thereby increasing their tolerance to high temperatures (Xia et al., 2009). In line with their findings, a study showed that cucumber treated with brassinosteroid exhibited increased GSH and AsA levels under HS, reducing oxidative damage (Calabrese & Mattson, 2011). Similar results were found that 24-epibrassinolide (EDR) treatment increased SOD, CAT activity and the levels of AsA and GSH in soybean under heat stress, indicating a crucial function of these enzymes in the detoxification of H2O2, O2.- and MDA levels (Ei et al., 2024).
These results suggest that 0.1% of P. guajava and A. sativum extracts are effective in protecting cucumbers from heat stress by improving their growth, water retention, photosynthetic activity and antioxidant protection. The changes suffered in cells or organisms at low doses of a stress condition reflect in environmentally induced altered phenotype, the above can be translated into quantitatively plasticity potential (Calabrese, 2014a; Calabrese, 2014b). It is this modest overcompensation response that is seen as the hermetic low-dose stimulation. The hormesis management can be a powerful strategy to satisfy the demand of the prevailing agriculture to maintain desirable yields in crops and increase the xenohormesis potential. But it is indispensable to consider that for the assessment and characterization of the hormesis process, the experimental designs require more doses, a greater simple population, and a heightened need for replication (Calabrese, 2014b). Future research should focus on improving application methods, evaluating long-term effects in the field and investigating potential antifungal capabilities under abiotic stress conditions.
CONCLUSIONS
This study showed that plant extracts, especially P. guajava, A. vera, A. sativum and M. sativa, significantly increased growth recovery, relative water content and tolerance to oxidative stress in cucumbers exposed to HS. The use of these extracts increased shoot fresh weight (27-41%) and plant height (12-26%) of cucumber, mitigating the negative effects of HS. However, in cereals, including maize, barley and rice, extract treatments did not result in significant effects, highlighting the crop-specific responses to the application of plant extracts. The use of these extracts improved enzymatic antioxidant activity and decreased levels of H2O2 and MDA, indicating their potential to alleviate oxidative damage on cucumber. Extracts from P. guajava and A. sativum reduced oxidative damage by decreasing the H2O2 and MDA while increasing the antioxidant enzymes, total chlorophyll and carotenoid content, and chlorophyll fluorescence Fv/Fm, suggesting accelerated recovery from stress. These results suggest that P. guajava and A. sativum extracts are effective in protecting cucumbers from HS by improving their growth, water retention, photosynthetic activity and antioxidant protection. Among the tested concentrations, 0.1% was most consistently effective in improving tolerance of HS, especially in cucumber, followed by cereals. Future research should focus on evaluating long-term effects in the field and investigating potential antifungal capabilities under abiotic stress conditions.




