INTRODUCTION
MATERIALS AND METHODS
D. longicolla isolates and plant material used
Comparison of D. longicolla inoculation methods
Disease parameters
Data analysis
RESULTS
Isolates and varieties effect on disease development
Inoculation method strongly influences disease development
Inoculation methods have an impact on pathogenicity and disease progress of Diaporthe longicolla
DISCUSSION
INTRODUCTION
Soybean (Glycine max L.) is one of the most important legume crop grown in the world. The crop is originated in East Asia 6000-9000 years ago and later introduced into different continents (Sedivy et al., 2017). According to USDA (2025) report, 422 million metric tons of soybean was harvested from all over the world in 2024/2025 production year. On the same year, 155,000 metric tons of soybean was produced in South Korea. Despite the increase in production, soybean crop is susceptible to more than 300 diseases, of which 35 are classified as economically important (Hosseini et al., 2023). Soybean diseases are categorized based on the time of infection into early-season, mid-season, and late-season diseases (Buzdar et al., 2025). Pod and stem blight disease caused by Diaporthe species is one of the most destructive disease in all over soybean growing areas.
D. longicolla (Hobbs) formerly known as Phomopsis longicolla Hobbs, a species within the genus D. Nitschke (previously Phomopsis [Sacc.] Bubák), is the main causal agent of pod and stem blight in soybean (Sureshbabu et al., 2025). This fungus is a seed-borne pathogen from species of ascomycete fungus in the family Diaporthaceae. Although it is primarily known as a seed-borne pathogen, it can be isolated from all parts of plant. Conidiomata of D. longicolla develop within large, effuse stromata and exhibit prominent beak-like structures. Alpha conidia are generated on phialidic conidiophores, which are often branched (Hobbs et al., 1985).
D. longicolla has been linked to diseases affecting seeds, pods, and stems. The pathogen traditionally known as a fungus lacking a teleomorph stage and producing pycnidia with long necks that release only alpha conidia, has recently been found to cause unusual stem symptoms, produce beta conidia, and form a teleomorph stage (Petrovic et al., 2018). The symptoms on the soybean stem begin with brown lesions that darken and elongate over time, leading to wilting of the upper stems and eventually plant death (Abdelmagid et al., 2022). In addition, zone lines (thin black lines) that did not create a distinct pattern can be developed inside the lower stem of mature soybean plants (Olson et al., 2015). This fungus persists as pycnidia and perithecia in soybean residue left in no-till fields, and as mycelium within infected seeds.
A major challenge in disease-resistant variety screening is the precise and rapid assessment of disease phenotypes, such as incidence rate, onset time, necrotic area, and disease index; which is essential for evaluating resistance effectively (Li et al., 2025). Inoculation methods have a significant impact on symptom development caused by Diaporthe sp. on soybean seedlings (Ghimire et al., 2019). Choosing the right method of pathogen inoculation is essential to study the resistance of the host plant towards particular pathogen. Researchers used different seedling inoculation techniques for the screening of soybean resistance varieties against Diaporthe sp. and to study the virulence of the isolates. Li (2018) mentioned cut-seedling inoculation as one of fastest method to screen variety resistance and have consistent initial inoculum across samples. In addition, Twizeyimana (2012) reported that the cut-stem inoculation technique was effective for evaluating M. phaseolina resistance in soybeans and could also enhance resistance screening in other crops like Phaseolus species. The toothpick inoculation method, which was used by different researchers, in both solid and liquid media is also considered one of the best method to screen resistance varieties. Keeling (1982) previously evaluated stem canker resistance by inserting toothpicks into 10-day-old soybean stems, but the method produced inconsistent disease responses across genotypes, leading to some susceptible varieties being mistakenly rated as resistant. Later, Campbell et al. (2017) modified toothpick inoculation method by combining pre-soaked toothpicks in liquid medium, D. aspalathi colonization on agar, inoculation above the cotyledon on three-week-old seedlings, sealing with petroleum jelly, and 72-hour high-humidity incubation; achieved high disease incidence in susceptible soybean lines. Moreover, Ghimire et al., (2019) tested various inoculation methods, including stem-wound method, toothpick method, mycelium contact method, and spore injection method to screen soybean resistance varieties against three Diaporthe sp., where he found comparatively lower disease severity on soybean plants inoculated with mycelium contact method and spore injection. Mena et al. (2019) also conducted an experiment using toothpick method, stem wounding, mycelium and ascospore inoculation, to evaluate the diversity of Diaporthe sp. Based on the results, the mycelium and ascospore suspension methods caused only slight stem browning, leading to the selection of the stem wounding method as the most effective (Mena et al., 2019).
In this study we conducted a comprehensive evaluation of eight inoculation methods on three- week-old soybean seedling. Widely cultivated soybean varieties in South Korea, Taekwang and Pyeongwon were inoculated with two virulent D. longicolla isolates using each technique under controlled environment and their susceptibility were evaluated based on their pathogenicity test result. Therefore, our study will provide an insight on how inoculation methods affect the development of pod and stem blight disease symptoms on soybean stem and contribute valuable information for future disease resistance screening and management practice in soybean cultivars.
MATERIALS AND METHODS
D. longicolla isolates and plant material used
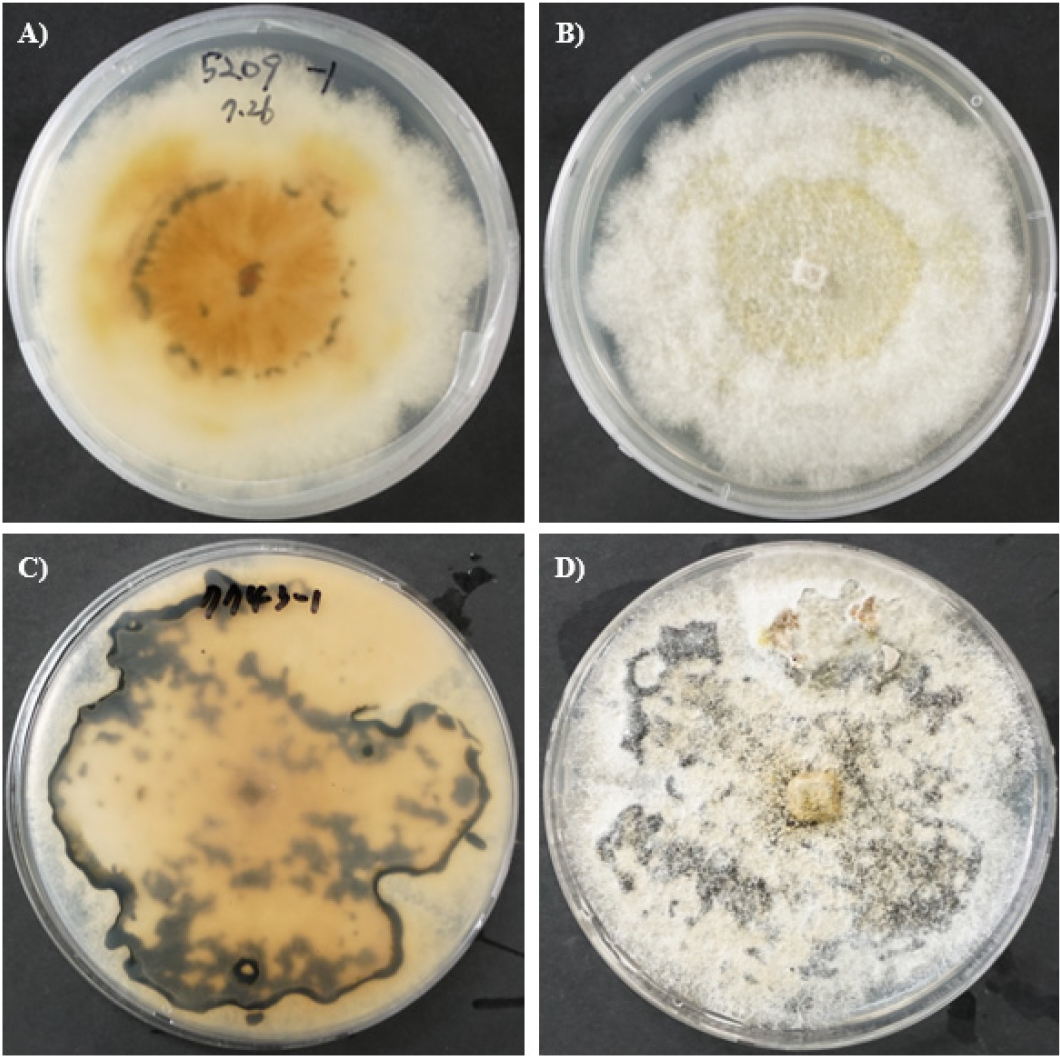
Two D. longicolla isolates, GJ52091 from Gimje and YC77131 from Yeoncheon, South Korea were used for this study (Fig. 1). These isolates were collected from infected soybean plants and isolated on water agar and then transferred to potato dextrose agar (PDA) media. The DNA of isolates GJ52091 and YC77131 were extracted and sequencing was conducted to confirm the pathogen. The ITS region sequences of both isolates were deposited in the GenBank database under accession numbers PX226042 and PX226092, respectively. After confirming the pathogen based on morphological and molecular characteristics, three individual plants of each of the two susceptible soybean varieties, Taekwang and Pyeongwon, were inoculated with the two isolates. These varieties are widely recognized for their susceptibility to a range of pathogens, including fungi, bacteria, virus, and nematodes, making them suitable hosts for evaluating disease responses in this study (Choi et al., 2023, Kang et al., 2015, Kang et al., 2021, Then et al., 2023).
Comparison of D. longicolla inoculation methods
In total, eight inoculation technique were tested on three- week-old soybean seedlings (Supplementary Fig. 1).
Mycelium plug inoculation technique was modified from (Li, 2018, Petrovic et al., 2023). Actively grown D. longicolla cultures were pressed with the open end of a 200 μL pipette tip to extract circular mycelium-agar plugs (0.5 mm in diameter). Three- week-old soybean seedlings were inoculated by cutting the stem apex 25mm above the first trifoliolate node using a sterile scissor. A pipette tip containing a mycelial plug was then immediately placed on each cut, with the mycelia oriented directly toward the exposed surface. Pipette tips were removed 2 days after inoculation.
For toothpick inoculation technique, sterile toothpicks were placed on fresh PDA plates containing D. longicolla culture (Campbell et al., 2017, Hosseini et al., 2024). After incubation for 21 days at 24 °C under a 12-hour photoperiod, the toothpicks overgrown with fungal mycelium and pycnidia were inserted into the plant stem at approximately a 45° angle and about 50 mm beneath the cotyledons. For the control group, a sterile toothpick was inserted into the soybean stem and all inoculated seedlings were sealed with petroleum jelly.
The third method of inoculation was spore injection. To prepare the inoculum, autoclaved flat toothpicks were placed on PDA plates containing D. longicolla culture, according to Ghimire et al. (2019). After 21 days of incubation, when the toothpicks were visibly colonized by the fungus and covered with pycnidia, they were transferred to approximately 100 mL of sterile distilled water and stirred with magnetic stirrer to remove the fungal structures from the toothpicks, then the resulting suspension was filtered through cheesecloth. A 1 mL aliquot of the spore suspension was injected into soybean stems approximately 50 mm below the cotyledon using a 3 mL disposable syringe fitted with a 22-gauge needle. Control plants were injected with sterile distilled water using the same method. In both inoculated and control plants, the injection site was sealed with petroleum jelly.
In addition, conidial suspension was also injected to three- week-old soybean seedlings. The conidial suspension was prepared from 21 days old D. longicolla culture, by washing off the fungal structure using 30 mL of sterile distilled water. The suspension was filtered by cheesecloth and injected into soybean seedlings using the same procedure mentioned above for spore injection.
Mycelium spraying inoculation technique was conducted by transferring five PDA plugs containing actively growing mycelium into 100 mL of potato dextrose broth and incubated in the dark under constant agitation at 120 rpm for 14 days. Following incubation, the harvested mycelium was suspended in approximately 30 mL of sterile distilled water and homogenized using an electric blender. The resulting suspension was then filtered through a cheesecloth to remove mycelial clumps, and the filtrate was adjusted to an optical density (OD) of 1 before spraying to soybean seedlings using airbrush atomizer. Control plants were sprayed with sterilized distilled water.
The stem wound technique was modified from Zhao et al. (2022). A thin slice approximately 5-7 mm in diameter was made along the stem, 2 cm below the cotyledon, using a sterile scalpel. Agar plugs containing actively growing mycelium from 14 day old cultures were carefully placed onto the wounded area. For control treatments, PDA plugs without the pathogen were similarly applied to wounded stems. In both cases, the wounds were sealed with petroleum jelly.
For mycelium contact method, 14-day-old mycelial plug approximately 5 mm in diameter was placed in contact with the stem of soybean plants, positioned about 50 mm below the cotyledon. For control treatments, PDA plugs without the pathogen were placed on the stem (Ghimire et al., 2019). In both cases, the plugs were wrapped with parafilm to prevent dehydration.
Ascospore inoculation method was conducted using Mena et al. (2019) method, with a little modification. To produce D. longicolla ascospore, plates containing mycelium plug with sterilized toothpicks were incubated for one month at 25℃ under 12hrs light/dark photoperiod. Colonized toothpicks were transferred to 30 mL of sterile distilled water to collect perithecia with the help of magnetic stirrer and then blended with electric blender. The resulting suspension was filtered through cheesecloth. A 10 μL drop of the spore suspension was then placed onto the unifoliate leaf petioles and on the stem. The control plants were inoculated with sterilized distilled water. Inoculated plants were immediately placed in a humid chamber and maintained for 48-hours post-inoculation.
Each experiment was conducted with three replicates and the entire experiment was repeated three independent times. Except for the ascospore inoculation tests, plants were placed under a small plastic house for two days to enhance humidity (Supplementary Fig. 2).
Disease parameters
Disease severity was measured using 0-5 scale (Supplementary Fig. 3) at 4, 7, 9, and 14 days after inoculation, where 0: no visible discoloration, 1: minor discoloration at the point of inoculation, 2: slight discoloration or small lesions measuring 1-2 mm, 3: necrotic lesions ranging from 2-5 mm, accompanied by some dark streaks on the stem, leaf wilting, and twisting, 4: lesions between 5-10 mm, with extensive necrosis, pronounced dark stem streaks, noticeable leaf and plant wilting, twisting, stunted growth, and occasional lodging, 5: lesions larger than 10 mm, with severe leaf necrosis, plant lodging, or death (Mathew et al., 2015). In addition, fourteen days after inoculation, measurements were taken for each plant to assess stem lesion length and overall stem length.
Data analysis
Based on the disease severity result, percent of disease severity index (DSI) was calculated using the formula described by Chiang et al. (2017), which is DSI (%) = [Σ (class frequency × score of rating class)] / [(total number of plants) × (maximal disease index)] × 100. In addition, to calculate AUDPC, the following formula described by Mena et al. (2019) was used. AUDPC =[(Yi+Yi+1)/2*(Xi+1-Xi)], where Yi = severity index according to infection index, Xi = times in days, and n = total observations number; where the final value of AUDPC is the sum of the areas by lapses, which result from the multiplication of the average reading of two consecutive dates (y value) by the lapse (days, x value) between readings.
All statistical analyses were performed in SAS 7.1. A two-factor factorial ANOVA was conducted to evaluate the effects of inoculation methods, varieties and isolates interaction. Post-hoc comparison was performed using Tukey’s Honest Significant Difference (HSD) for pairwise comparison. The significant threshold was set at α = 0.05.
RESULTS
Isolates and varieties effect on disease development
Soybean seedlings were monitored daily to observe the disease onset after inoculation. Based on the evaluation, ‘Pyeongwon’ variety showed faster symptom development, compared to ‘Taekwang’ variety. In addition, isolate GJ52091 induced earlier disease symptoms compared to isolate YC77431. At 14 days after inoculation (DAI), stem lesion length were measured and significant difference was observed between isolates. The D. longicolla isolate GJ52091 caused the longest stem lesion, which was 5.1 cm, while isolate YC77431 caused the shortest stem lesion measured 0.3 cm (Table 1). In addition, highest mean lesion length was recorded on both varieties inoculated with isolate GJ52091, compared with isolate YC77431 (Table 2). Furthermore, disease incidence and severity also showed variation among samples. Disease severity was monitored starting at 4 DAI, and continued for fourteen days. The highest disease severity was recorded on ‘Taekwang’ variety inoculated with isolate GJ52091, compared with ‘Pyeongwon’ variety (Fig. 3).
Table 1.
Mean stem lesion length of soybean seedlings inoculated with D. longicolla isolates using eight inoculation techniques.
Table 2.
Summary on average lesion lengths among isolates and varieties.
| Isolates | Varieties | Mean lesion Length |
| GJ5209 | Taekwang | 2.53a |
| Pyeongwon | 2.2ab | |
| YC7743 | Taekwang | 2b |
| Pyeongwon | 1.83b |
Inoculation method strongly influences disease development
A total of eight inoculation methods were evaluated using two soybean varieties inoculated with two virulent D. longicolla isolates. The result of different inoculation methods of D. longicolla on soybean seedlings showed significant variation in days to disease onset, where cut-seedling, toothpick and stem wound inoculation methods consistently induced the earliest disease onset, which was 2 DAI on both soybean cultivars and isolates. In contrast, spraying of mycelium and mycelium contact inoculation methods led to delayed symptom development. Soybean seedlings inoculated with ascospore didn’t show any symptoms of blight.
On the other hand, a two-factor ANOVA revealed a highly significant effect of inoculation method on stem lesion length (p = 0.0072). It showed that cut-seedling inoculation produced significantly longer lesion than all other methods (4.25 cm), while mycelial spray and ascospore inoculation didn’t form any lesion (Table 3).
Table 3.
Comparison of mean lesion lengths among different inoculation methods.
| Inoculation Method | Mean Lesion Length |
| Cut-seedling | 4.25a |
| Stem wound | 2.85b |
| Toothpick | 2.48b |
| Conidial suspension | 1.68c |
| Mycelial contact | 1.18c |
| Spore injection | 1.18c |
Inoculation methods have an impact on pathogenicity and disease progress of Diaporthe longicolla
A significant difference on disease severity and disease progression was observed between isolates inoculated with the eight inoculation methods tested on this study. Disease severity was monitored starting at four days after inoculation, and continued for fourteen days. The highest disease severity percentage recorded 14 DAI was 100%, which was on ‘Taekwang’ variety inoculated with isolate GJ52091 using toothpick inoculation and on both varieties inoculated with isolate GJ52091 using cut-seedling inoculation method. In addition, plant death was observed when soybean varieties were inoculated with isolate GJ52091 using cut-seedling and toothpick inoculation methods (Fig. 2). In contrast, the lowest disease severity (6.67%) was observed on seedlings inoculated with isolate YC77431 via mycelium spraying. Ascospore of both isolates inoculated on ‘Taekwang’ and ‘Pyeongwon’ varieties didn’t show any disease symptoms (Fig. 3).

Fig. 2.
Eight methods of inoculating D. longicolla on three- week-old soybean seedlings: (A) toothpick inoculation, (B) stem-wound inoculation, (C) mycelium contact, (D) cut-seedling inoculation, (E) spore injection, (F) conidial suspension injection, (G) spraying of mycelium, and (H) ascospore inoculation. The inoculated plants (on the left side of each category) were compared with the control plants (on the right side of each category) 14 days after inoculation (DAI).
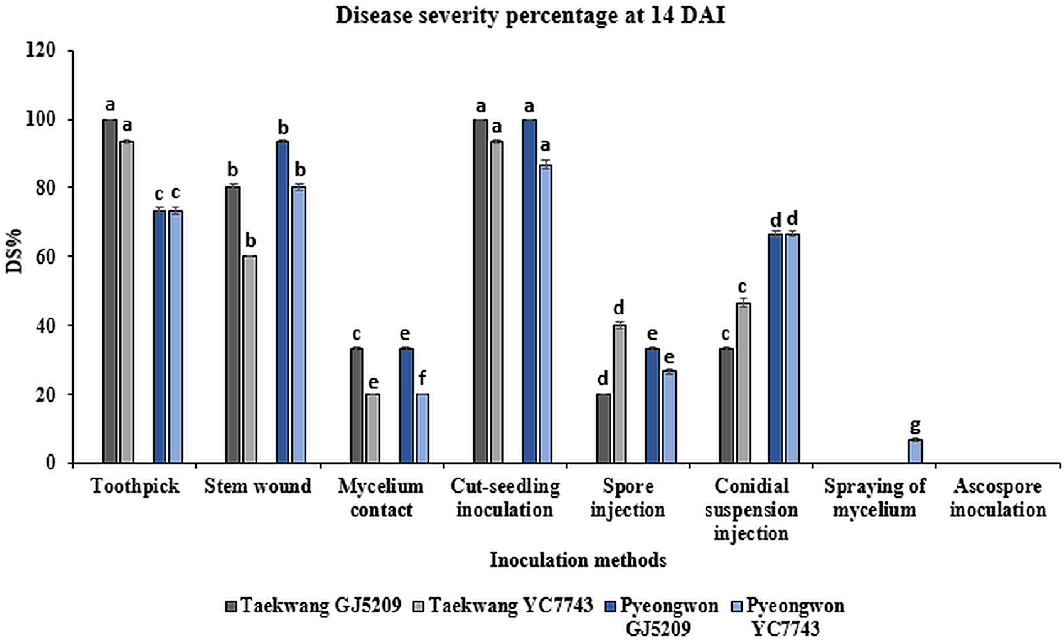
Fig. 3.
Disease severity percentages in soybean seedlings inoculated with D. longicolla isolates using various methods, calculated 14 DAI. The disease severity was measured using a 0-5 scale, and the control group exhibited no detectable symptoms (value=0). Different letters on different bars indicate significant at p < 0.0001.
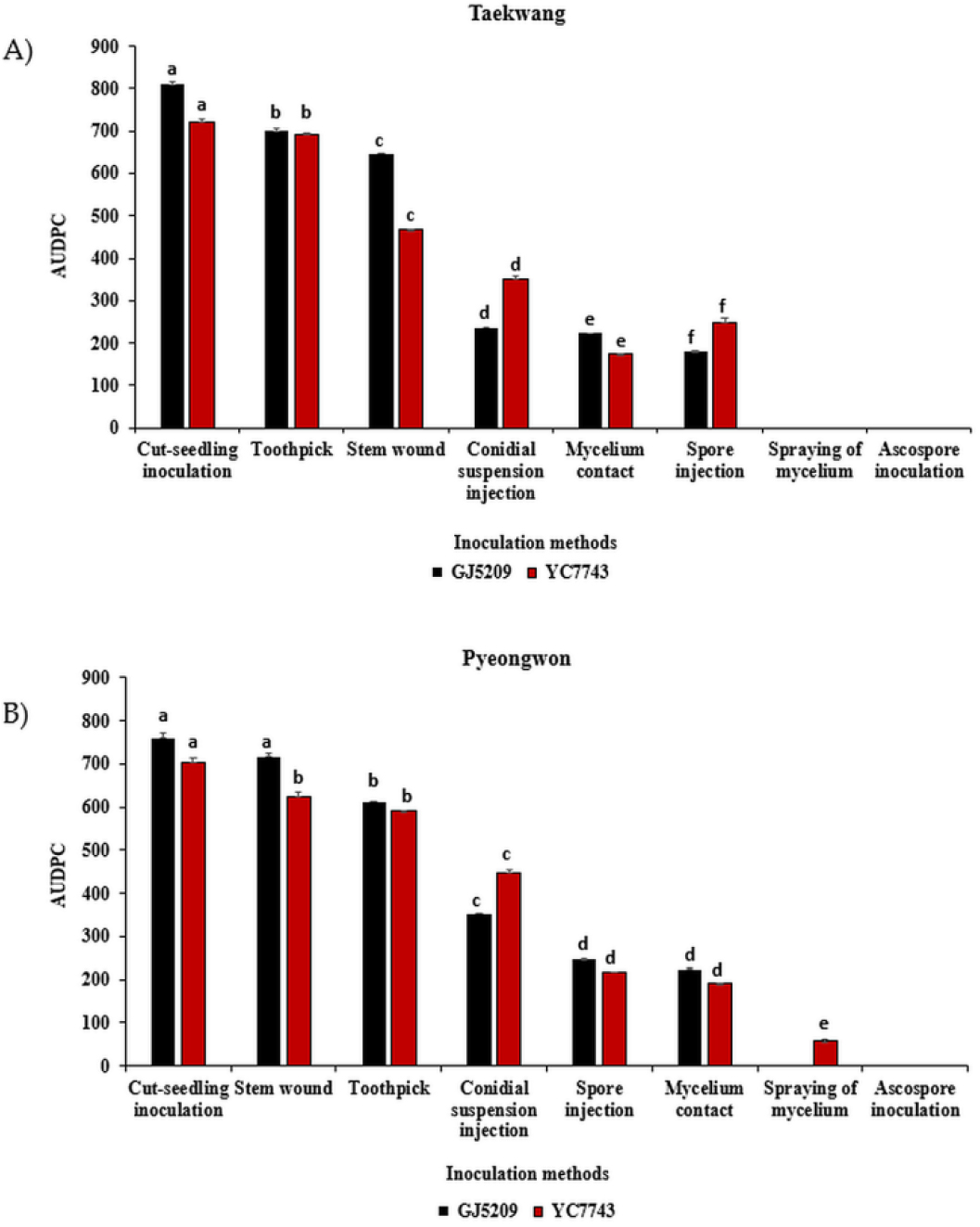
The AUDPC was used to evaluate the progression of pod and stem blight disease in two soybean varieties inoculated with two D. longicolla isolates and across the eight inoculation methods. In general, isolate GJ52091 caused more severe disease symptoms than isolate YC77431, as reflected by higher mean AUDPC values across all treatments. However, isolate YC77431 inoculated on both varieties using conidial suspension injection method showed higher AUDPC compared with isolate GJ52091 (Fig. 4, Supplementary Table 1).
Among the inoculation methods, cut-seedling inoculation resulted in the highest AUDPC values on both isolates and varieties, particularly on Variety ‘Taekwang’ inoculated with isolate GJ52091 (AUDPC = 810). However, depending on the variety, some inoculation methods produced different responses among the tested samples. In ‘Taekwang’, cut-seedling inoculation was followed by toothpick and then stem-wound inoculation, whereas in ‘Pyeongwon’ it was followed by stem-wound and then toothpick inoculation. In contrast, ascospore and mycelium spraying produced the lowest AUDPC values, on both varieties and isolates (Fig. 4, Supplementary Table 1).
DISCUSSION
Plant pathogen inoculation is a fundamental method in plant disease research, used to assess pathogen virulence, study infection processes, and support resistance breeding efforts (Shen et al., 2024). To evaluate soybean seedlings for resistance and determine the aggressiveness of Diaporthe sp., researchers have developed various early season artificial inoculation techniques, emphasizing the need for an appropriate method to accurately assess the resistance of different varieties to stem and pod blight disease. Seedling inoculation techniques offers major advantages by enabling accurate pod and stem blight disease resistance screening within 2-3 weeks, far faster than traditional end-of-season seed assessments that require 102-164 days (Li, 2018). These assays also allow rapid evaluation of host response, enabling earlier selection and reducing the time required to identify resistant genotypes. In addition, young plants provide a more uniform and easily controlled system, ensuring consistent inoculum placement and reducing environmental variability across treatments. Seedling inoculation also requires fewer resources, allowing large populations to be screened efficiently in greenhouse or growth-chamber conditions. Because many defense responses to Diaporthe spp. are expressed early in development, seedling tests often provide a reliable indication of mature-plant resistance. Therefore, choosing effective invitro seedling assay is important to ensure infection and identify strong and stable resistant genotype.
Our study demonstrated variability in disease severity among the eight inoculation methods, with cut-seedling, toothpick and stem-wound inoculation methods resulting in significant levels of lesion development (Fig. 2). Many researchers also confirmed the effectiveness of these inoculation methods. Shuxian (2011) mentioned the cut-seedling assay method as an effective method to evaluate isolates and their quantitative measurements of virulence on soybean plants. In addition, (Li, 2018) performed the cut-seedling inoculation technique to screen soybean resistant varieties against D. longicolla and reported large stem lesion length and AUDPC on soybean seedling. The research conducted by Petrovic et al. (2023) indicated that the malondialdehyde (MDA) accumulation, which is an indicator of oxidative stress in plants were higher in cut-seedling inoculated and toothpick inoculated soybean cultivars, compared to the plants inoculated with mycelial contact inoculation technique. Moreover, the results of various inoculation methods evaluated by Ghimire et al. (2019) indicated that toothpick and stem-wound inoculation methods are effective techniques to evaluate the virulence of different Diaporthe species compared with mycelium contact and spore injection methods. Campbell et al. (2017) also proved toothpick inoculation method as an effective technique after testing D. aspalathi isolates on seven susceptible soybean cultivars. The similarity of inoculation methods mentioned above, cut-seedling, toothpick, and stem-wound techniques is that they all involve wounding of the host plant. These wounds provide entry points for the pathogen, allowing it to invade and colonize the vascular tissues. In addition, sealing the inoculated area helps retain moisture, which enhances fungal growth and accelerates disease development.
In contrast, mycelium contact, spore injection and conidial suspension produced average inoculation outcomes and induce stem lesion formation, but compared with the above mentioned inoculation techniques, the severity is less. Additionally, mycelial spraying and ascospore inoculation didn’t show any difference. Both varieties inoculated with ascospore and ‘Taekwang’ variety sprayed with mycelium didn’t show any disease symptom. These results are consistent with Mena et al. (2019), where the mycelium and ascospore suspension resulted in only slight stem browning with no formation of typical lesions. Therefore, in the absence of wounding, surface contact or spraying of the pathogen result in low or no disease development.
High interaction effects were observed between isolate, variety, and inoculation method, suggesting that disease progression was influenced not only by isolate virulence and host susceptibility but also by the mode of pathogen delivery. In this study, disease symptoms were first observed at 2 DAI in the most destructive combinations, which was ‘Taekwang’ inoculated with isolate GJ52091 using cut-seedling inoculation method, and peaked between 7 and 14 DAI. Disease severity and AUDPC was also higher in this combination.
The overall result indicate that cut-seedling inoculation method is the most effective and rapid approach, providing an efficient and reliable method for soybean variety screening. This inoculation method ensures an even distribution of fungal inoculum among treatments, thereby minimizing bias and enhancing result reliability. In general, this study provides important insights into how different inoculation methods influence the development and severity of stem and pod blight diseases in soybean seedlings, enhancing our understanding of pathogen virulence and contributing to the improvement of disease evaluation techniques in soybean research.